
Monthly Archives: July 2022
Cartoon – Your Integrity is on the Line
Importance of Leadership Integrity
R-E-S-P-E-C-T
Importance of Showing Respect
SEC may take ‘fresh look’ at auditor conflicts of interest: Gensler

Dive Brief:
- Securities and Exchange Commission (SEC) Chair Gary Gensler has asked the Public Company Accounting Oversight Board (PCAOB) Chair Erica Williams to consider adding auditor independence standards to its agenda and the SEC itself may need to take a “fresh look” at its own rules on the independence issue, Gensler said during a webinar Wednesday.
- SEC staff has seen “situations of decreased vigilance” when it comes to auditor independence and Gensler expressed concern that conflicts of interest stemming from auditors and affiliated firms serving the same client persist 20 years after the Sarbanes Oxley Act directed the SEC to take steps to create stronger barriers between auditors and other parts of their firms, he said.
- “A number of firms spun out their consulting businesses in the days shortly before and after Sarbanes-Oxley. Over the past 20 years, however, many of these firms went on to rebuild them again. PCAOB inspections continue to identify independence — and lack of professional skepticism — as perennial problem areas,” Gensler said.
Dive Insight:
Gensler spoke during a webinar hosted by the Center for Audit Quality commemorating the 20th anniversary Sarbanes-Oxley Act, which established the PCAOB in the wake of the Enron and WorldCom accounting scandals to oversee accounting firms that audit public companies.
The SEC chair has been shaking up the oversight of auditors for some time, ousting William Duhnke as PCAOB chair last year. Last fall SEC Acting Chief Accountant Paul Munter underscored the importance of independent audits as an investor safeguard, saying that an auditor that provides extensive non-audit services to an entity that has an active mergers and acquisitions business model must continually monitor the impacts of all such transitions on its audit engagement to ensure that the auditor remains independent of all of its audit clients.
During the talk Gensler was also critical of the sluggish pace at which PCAOB has undertaken the responsibility that it was given to update interim standards that it inherited from the American Institute of Certified Public Accountants.
“Historically … the PCAOB has been too slow to update auditing standards. Twenty years later, most of those interim standards remain,” Gensler said. But he expressed confidence that Williams, who took over as chair of the U.S. audit watchdog in January, and the board would “live up to Congress’s original vision with respect to standard-setting. I hope we can make some progress before Sarbanes-Oxley can legally drink.”
In May the PCAOB announced plans to update almost all of the remaining interim standards.
San Francisco, New York state call monkeypox an emergency: 5 updates
New York state declared an imminent threat and San Francisco issued a state of emergency over monkeypox July 28 as the virus continues to spread in the U.S., NBC News reported.
The news comes after the World Health Organization declared monkeypox a global emergency July 23 and as the CDC reported 4,907 confirmed cases nationwide as of July 28. California and New York account for more than 40 percent of the reported cases in the U.S., according to The Washington Post.
In a statement, New York State Commissioner of Health Mary Bassett, MD, said the declaration allows local health departments “to access additional state reimbursement, after other federal and state funding sources are maximized, to protect all New Yorkers and ultimately limit the spread of monkeypox in our communities.” It covers monkeypox prevention response and activities from June 1 through the end of the year.
In San Francisco, the monkeypox public health emergency takes effect Aug. 1, city officials said in a news release. The release, from Mayor London Breed and the San Francisco Department of Public Health, said the declaration “will mobilize city resources, accelerate emergency planning, streamline staffing, coordinate agencies across the city, allow for future reimbursement by the state and federal governments and raise awareness throughout San Francisco about [monkeypox].”
Four other updates:
1. HHS announced July 28 that nearly 800,000 additional monkeypox vaccine doses will be available for distribution to states and jurisdictions. The 786,000 additional doses are on top of the more than 300,000 doses already distributed. This means the U.S. has secured a total of about 1.1 million doses “that will be in the hands of those who need them in the next several weeks,” HHS Secretary Xavier Becerra said during a July 28 news conference. The additional doses will be allocated based on the total population of at-risk people and the number of new cases in each jurisdiction. “This strategy ensures that jurisdictions have the doses needed to complete the second dose of this two-dose vaccine regimen for those who have been vaccinated over the past month,” HHS said in a news release.
2. As of the morning of July 29, the U.S. has held off on declaring a national monkeypox emergency. Mr. Becerra said July 28 that HHS “continue[s] to monitor the response throughout the country on monkeypox” and will weigh any decision regarding a public health emergency declaration based on the response.
3. The monkeypox response is straining public health workers. Health experts are concerned over how the monkeypox response will further deplete the nation’s public health workforce, still strained and burnt out from the ongoing COVID-19 pandemic. Barriers to testing, treatment and vaccine access largely mirror the missteps in the early coronavirus response, Megan Ranney, MD, emergency physician and academic dean of Brown University School of Public Health in Providence, R.I, told The Washington Post. “I can’t help but wonder if part of the delay is that our public health workforce is so burned out,” she said. “Everyone who’s available to work on epidemiology or contract tracing is already doing it for COVID-19.”
4. Monkeypox testing demand is low, commercial laboratories told CNN. In recent weeks, five major commercial laboratories have begun monkeypox testing, giving the nation capacity to conduct 80,000 tests per week. While Mayo Clinic Laboratories can process 1,000 samples a week, it’s received just 45 specimens from physicians since it began monkeypox testing July 11, according to the July 28 CNN report. “Without testing, you’re flying blind,” William Morice, MD, PhD, president of Mayo’s lab and chair of the board of directors at the American Clinical Laboratory Association, told the news outlet. “The biggest concern is that you’re not going to identify cases and [monkeypox] could become an endemic illness in this country. That’s something we really have to worry about.”
Critics say Mark Cuban’s pharmacy isn’t tackling the big issue: brand-name drugs

Mark Cuban’s pharmacy, Cost Plus Drug Co., has hundreds of drugs marked at discounted prices, but some pharmacy experts say there’s a larger problem that needs fixing, CNBC reported July 28.
The online pharmacy launched in January with about 100 drugs, and by its one-year anniversary, plans to have more than 1,500 medications, according to the company’s website. The business model, which allocates for a $3 pharmacy dispensing fee, $5 shipping fee and a 15 percent profit margin with each order, aims to uproot the pharmaceutical industry, which has faced criticism for years about its opaque business practices.
Gabriel Levitt, the president of PharmacyChecker, a company that monitors the cheapest drug prices, told CNBC there’s more to be done.
“As much as I support the venture, what they’re doing does not address the big elephant in the room,” Mr. Levitt said. “It’s really brand-name drugs that are increasing in price every year and forcing millions of Americans to cut back on medications or not take them at all.”
Brand-name drugs are 80 percent to 85 percent more expensive than generics since brand-name drugs have to repeat clinical tests to prove efficacy, according to the FDA. Cost Plus Drug Co. only offers generics. Mr. Cuban told CNBC he hopes to sell brand-name medications “within six months,” but added that it’s a tentative timeline.
Developing a compelling value proposition for employees
https://mailchi.mp/ff342c47fa9e/the-weekly-gist-july-22-13699925?e=d1e747d2d8
As we’ve been discussing, the COVID pandemic and ensuing economic environment have driven health system job vacancies and attrition rates to all-time highs. Right now, for myriad reasons, many hospital workers are deciding that the financial, emotional, and professional benefits of working for a hospital are outweighed by the toll working in a hospital takes on them personally.
Health systems are responding to this challenge with a wide variety of discrete measures—including hiring and retention bonuses, incentive pay, employee wellbeing initiatives, and expanded professional development opportunities— that target specific groups of employees, but don’t form a long-term solution to workforce instability.
To rebuild a stable and committed workforce, health systems must create, and then communicate, a compelling employee value proposition—a concise statement highlighting why employees should work for them.
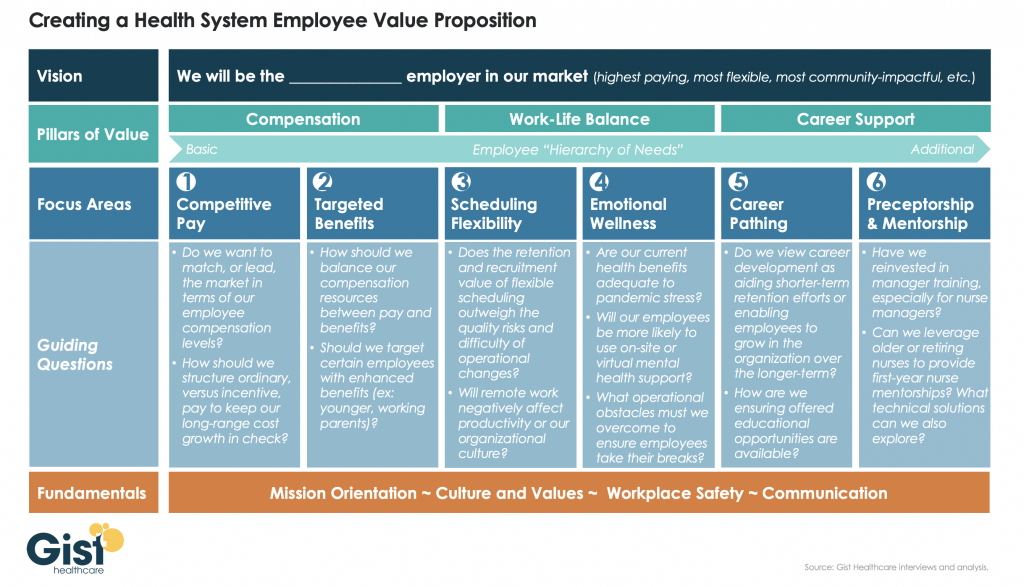
The graphic above shows what we believe are the key components of a successful employee value proposition, which must have a clear vision and focus on the things most important to employee needs: compensation, work-life balance, and career support. Systems can use the guiding questions listed in each column to craft a value proposition that is differentiated in their local labor market, informed by their level of resources, and undergirded by their own culture and values.
Value-based care isn’t yielding much “value.”
https://mailchi.mp/ff342c47fa9e/the-weekly-gist-july-22-13699925?e=d1e747d2d8

Despite the hype, accountable care organizations (ACOs) and other Medicare-driven payment reform programs intended to improve quality and lower healthcare spending haven’t bent the cost curve to the extent many had hoped.
A recent and provocative opinion piece in STAT News, from health policy researcher Kip Sullivan and two single-payer healthcare advocates, calls for pressing pause on value-based payment experimentation. The authors argue that current attempts to pay for value have ill-defined goals and hard-to-measure quality metrics that incentivize reducing care and upcoding, rather than improving outcomes.
The Gist: We agree with the authors that current value-based care experiments have been disappointing.
The intention is good, but the execution has been bogged down by entrenched industry dynamics and slow-to-move incumbents. One fair criticism: ACOs and other “total cost management” reforms largely focus on the wrong problem. They address utilization, rather than excessive price.
But we’re having a price problem in the US, not a utilization problem. Europeans, for example, have more physician visits each year than Americans, yet spend less per-person on healthcare. It’s our high prices—for everything from physician visits to hospital stays to prescription drugs—that drive high healthcare spending.
The root cause: our third-party payer structure actively discourages real efforts to lower price—every player in the value chain, including providers, brokers, and insurers, does better economically as prices increase. That’s why price control measures like reference pricing or price caps have been nonstarters among industry participants.
Recent reforms that increase price transparency, while not the entire solution, at least shine a light on the real challenges our healthcare system faces.





