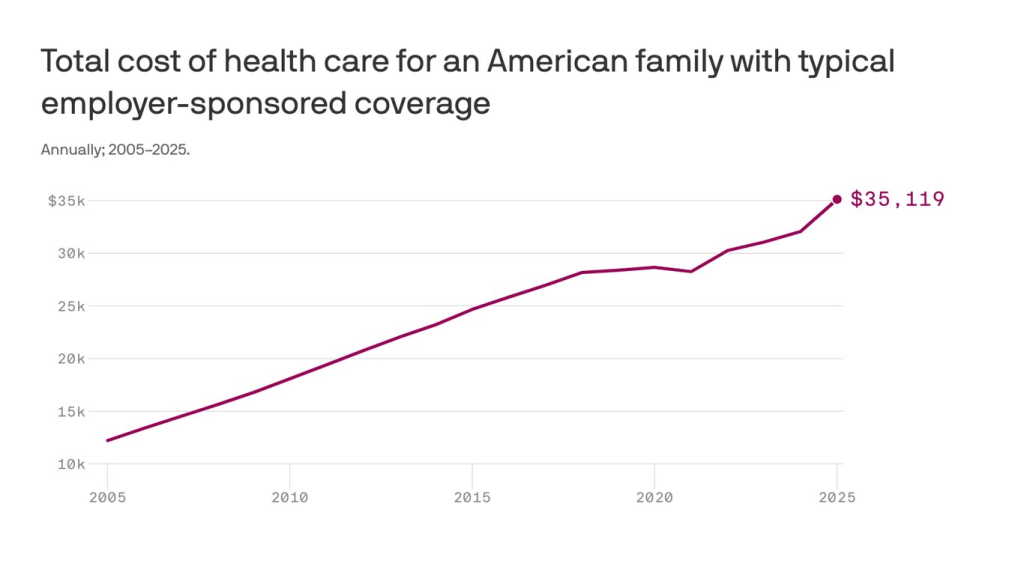President Donald Trump has made big promises about fixing American healthcare. Now comes the moment that separates talk from action.
With the 2026 midterms fast approaching and congressional attention soon shifting to electoral strategy, the window for legislative results is closing quickly. This summer will determine whether the administration turns promises into policy or lets the opportunity slip away.
Trump and his handpicked healthcare leaders — HHS Secretary Robert F. Kennedy Jr. and FDA Commissioner Dr. Marty Makary — have identified three major priorities: lowering drug prices, reversing chronic disease and unleashing generative AI. Each one, if achieved, would save tens of thousands of lives and reduce costs.
But promises are easy. Real change requires political will and congressional action. Here are three tests that Americans can use to gauge whether the Trump administration succeeds or fails in delivering on its healthcare agenda.
Test No. 1: Have Drug Prices Come Down?
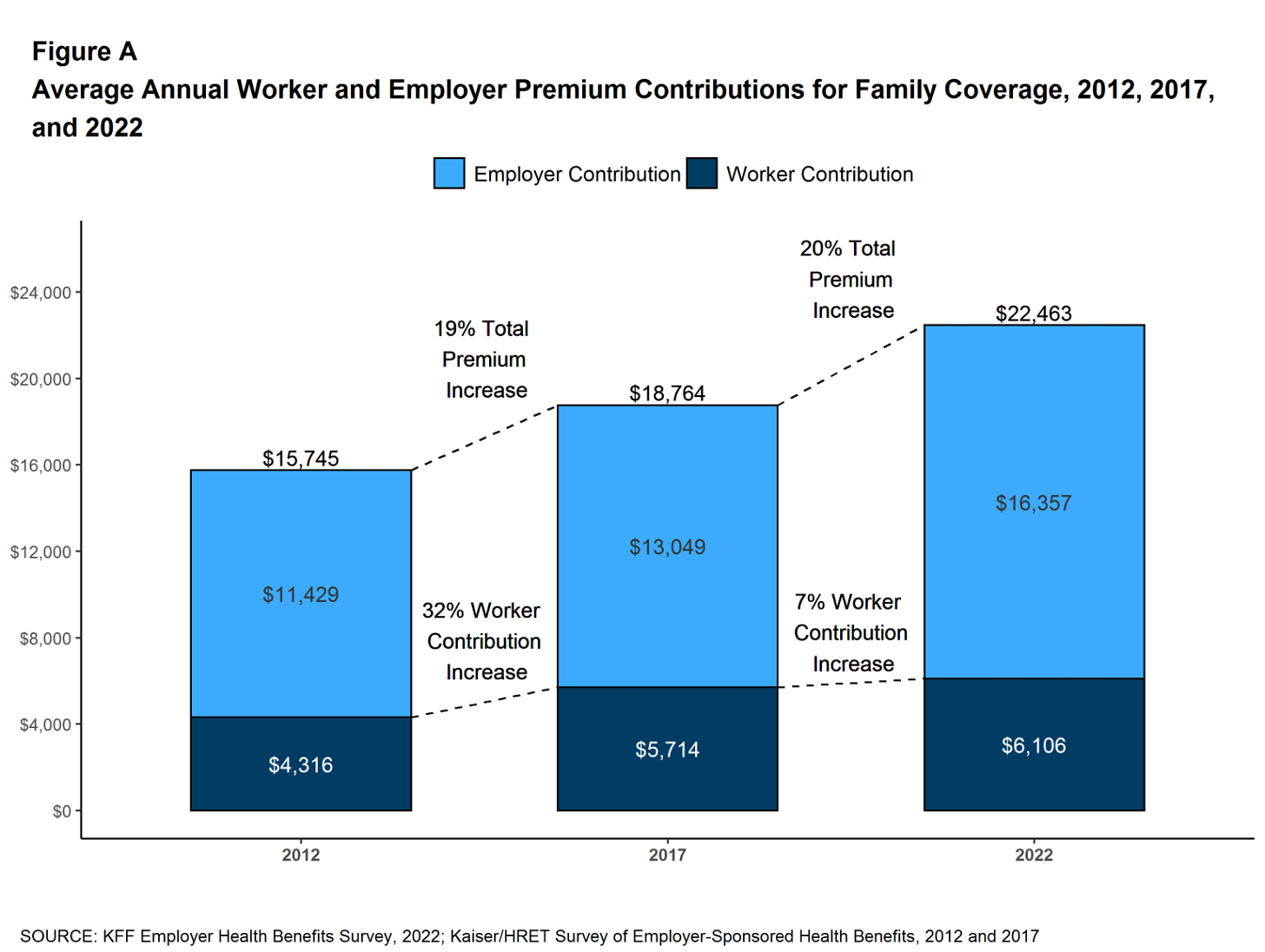
Americans pay two to four times more for prescription drugs than citizens in other wealthy nations. This price gap has persisted for more than 20 years and continues to widen as pharmaceutical companies launch new medications with average list prices exceeding $370,000 per year.
One key reason for the disparity is a 2003 law that prohibits Medicare from negotiating prices directly with drug manufacturers. Although the Inflation Reduction Act of 2022 granted limited negotiation rights, the initial round of price reductions did little to close the gap with other high-income nations.
President Trump has repeatedly promised to change that. In his first term, and again in May 2025, he condemned foreign “free riders,” promising, “The United States will no longer subsidize the healthcare of foreign countries and will no longer tolerate profiteering and price gouging.”
To support these commitments, the president signed an executive order titled “Delivering Most-Favored-Nation (MFN) Prescription Drug Pricing to American Patients.” The order directs HHS to develop and communicate MFN price targets to pharmaceutical manufacturers, with the hope that they will voluntarily align U.S. drug prices with those in other developed nations. Should manufacturers fail to make significant progress toward these targets, the administration said it plans to pursue additional measures, such as facilitating drug importation and imposing tariffs. However, implementing these measures will most likely require congressional legislation and will encounter substantial legal and political challenges.
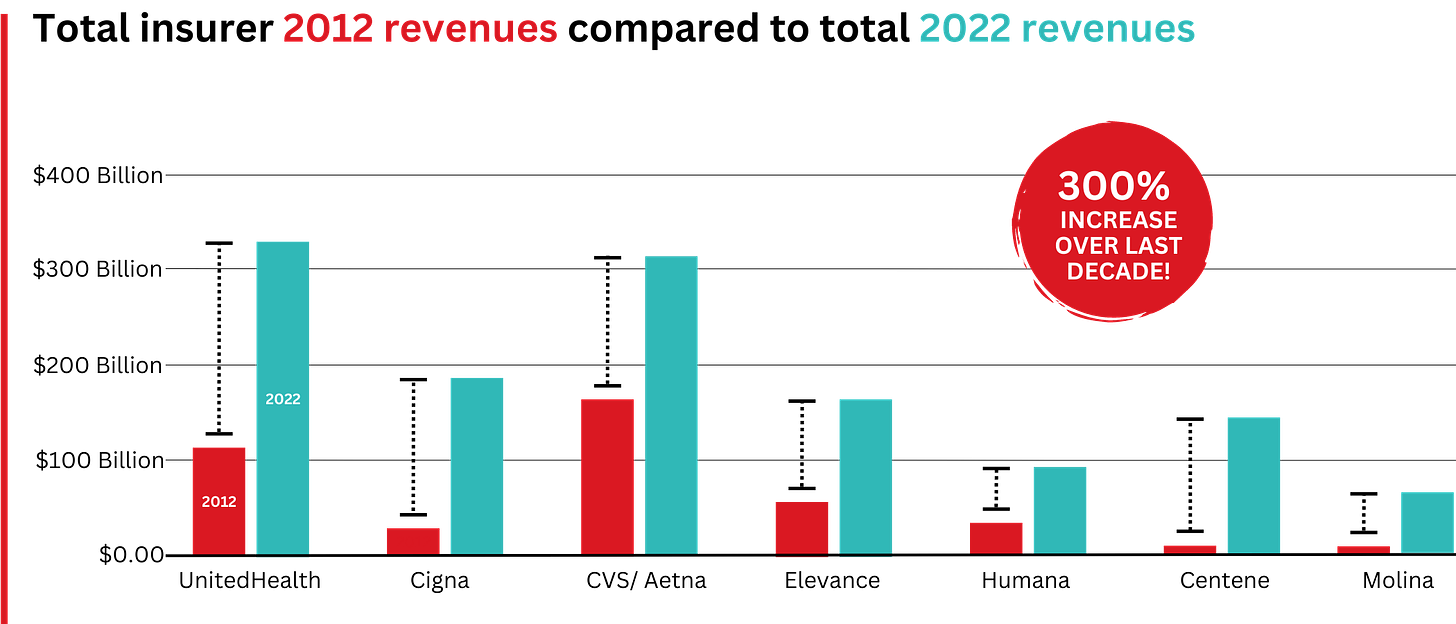
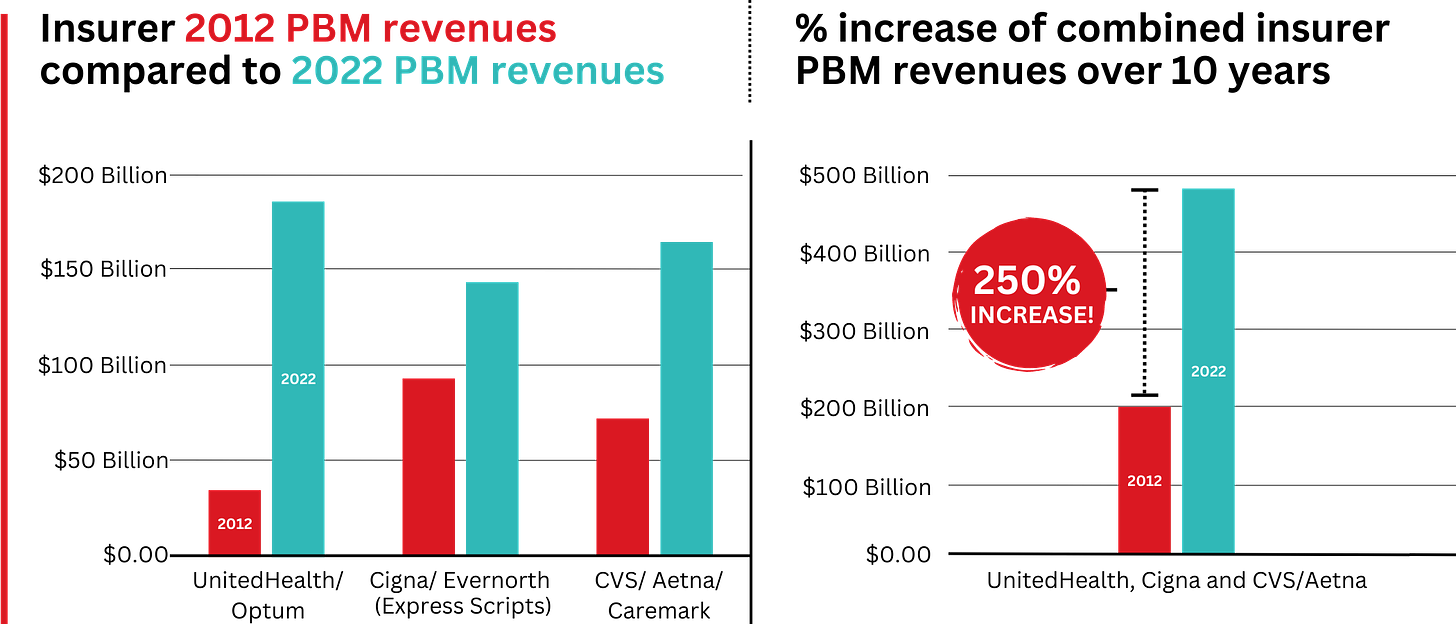
The pharmaceutical industry knows that without congressional action, there is no way for the president to force them to lower prices. And they are likely to continue to appeal to Americans by arguing that lower prices will restrict innovation and lifesaving drug development.
But the truth about drug “innovation” is in the numbers: According to a study by America’s Health Insurance Plans, seven out of 10 of the largest pharmaceutical companies spend more on sales and marketing than on research and development. And if drugmakers want to invest more in R&D, they can start by requiring peer nations to pay their fair share — rather than depending so heavily on U.S. patients to foot the bill.
If Congress fails to act, the president has other tools at his disposal. One effective step would be for the FDA to redefine “drug shortages” to include medications priced beyond the reach of most Americans. That change would enable compounding pharmacies to produce lower-cost alternatives just as they did recently with GLP-1 weight-loss injections.
If no action is taken, however, and Americans continue paying more than twice as much as citizens in other wealthy nations, the administration will fail this crucial test.
Test No. 2: Did Food Health, Quality Improve?
Obesity has become a leading health threat in the United States, surpassing smoking and opioid addiction as a cause of death.
Since 1980, adult obesity rates have surged from 15% to over 40%, contributing significantly to chronic diseases, including type 2 diabetes, heart disease and multiple types of cancers.
A major driver of this epidemic is the widespread consumption of ultra-processed foods: products high in added sugar, unhealthy fats and artificial additives. These foods are engineered to be hyper-palatable and calorie-dense, promoting overconsumption and, in some cases, addictive eating behaviors.
RFK Jr. has publicly condemned artificial additives as “poison” and spotlighted their impact on children’s health. In May 2025, he led the release of the White House’s Make America Healthy Again (MAHA) report, which identifies ultra-processed foods, chemical exposures, lack of exercise and excessive prescription drug use as primary contributors to America’s chronic disease epidemic.
But while the report raises valid concerns, it has yet to produce concrete reforms.
To move from rhetoric to results, the administration will need to implement tangible policies.
Here are three approaches (from least difficult to most) that, if enacted, would signify meaningful progress:
- Front-of-package labeling. Implement clear and aggressive labeling to inform consumers about the nutritional content of food products, using symbols to indicate healthy versus unhealthy options.
- Taxation and subsidization. Impose taxes on unhealthy food items and use the revenue to subsidize healthier food options, especially for socio-economically disadvantaged populations.
- Regulation of food composition. Restrict the use of harmful additives and limit the total amount of fat and sugar included, particularly for foods aimed at kids.
These measures will doubtlessly face fierce opposition from the food and agriculture industries. But if the Trump administration and Congress manage to enact even one of these options — or an equivalent reform — they can claim success.
If, instead, they preserve the status quo, leaving Americans to decipher nutritional fine print on the back of the box, obesity will continue to rise, and the administration will have failed.
Test No. 3: Are Patients Using Generative AI To Improve Health?
The Trump administration has signaled a strong commitment to using generative AI across various industries, including healthcare. At the AI Action Summit in Paris, Vice President JD Vance made the administration’s agenda clear: “I’m not here this morning to talk about AI safety … I’m here to talk about AI opportunity.”
FDA Commissioner Dr. Marty Makary has echoed that message with internal action. After an AI-assisted scientific review pilot program, he announced plans to integrate generative AI across all FDA centers by June 30.
But internal efficiency alone won’t improve the nation’s health. The real test is whether the administration will help develop and approve GenAI tools that expand clinical access, improve outcomes and reduce costs.
To these ends, generative AI holds enormous promise:
- Managing chronic disease: By analyzing real-time data from wearables, GenAI can empower patients to better control their blood pressure, blood sugar and heart failure. Instead of waiting months between doctor visits for a checkup, patients could receive personalized analyzes of their data, recommendations for medication adjustments and warnings about potential risk in real time.
- Improving diagnoses: AI can identify clinical patterns missed by humans, reducing the 400,000 deaths each year caused by misdiagnoses.
- Personalizing treatment: Using patient history and genetics, GenAI can help physicians tailor care to individual needs, improving outcomes and reducing side effects.
These breakthroughs aren’t theoretical. They’re achievable. But they won’t happen unless federal leaders facilitate broad adoption.
That will require investing in innovation. The NIH must provide funding for next-generation GenAI tools designed for patient empowerment, and the FDA will need to facilitate approval for broad implementation. That will require modernizing current regulations. The FDA’s approval process wasn’t built for probabilistic AI models that rely on continuous application training and include patient-provided prompts. Americans need a new, fit-for-purpose framework that protects patients without paralyzing progress.
Most important, federal leaders must abandon the illusion of zero risk. If American healthcare were delivering superior clinical outcomes, managing chronic disease effectively and keeping patients safe, that would be one thing. But medical care in the United States is far from that reality. Hundreds of thousands of Americans die annually from poorly controlled chronic diseases, medical errors and misdiagnoses.
If generative AI technology remains confined to billing support and back-office automation, the opportunity to transform American healthcare will be lost. And the administration will have failed to deliver on this promise.
When I teach strategy at Stanford’s Graduate School of Business, I tell students that the best leaders focus on a few high-priority goals with clear definitions of success — and a refusal to accept failure. Based on the administration’s own words, grading the administration on these three healthcare tests will fulfill those criteria.
However, with Labor Day just months away, the window for action will soon close. The time for presidential action is now.