
Category Archives: Decision Making
Cartoon – A Good Investment
Is our collaborative culture slowing down our ability to act quickly?
https://mailchi.mp/cd8b8b492027/the-weekly-gist-january-26-2024?e=d1e747d2d8

“We have a collaborative culture; it’s one of our system’s core values. But it takes us far too long to make decisions.” A health system CEO made this comment at a recent meeting, giving voice to a dilemma many system executives are no doubt facing. Of course, leaders want their teams to collaborate—in any important decision, we want to hear different voices, consider diverse points of view, and incorporate various areas of expertise.
On the other hand, collaboration takes time, which we don’t have right now. It also can add complexity, be the enemy of clear direction, and muddy accountability. This CEO went on to make an essential connection: “My concern is that this protracted decision making isn’t just a process problem, but that it’s showing up in our results.
Take performance improvement—we all quickly agreed we need to cut costs, but it’s taking far too long for us to act, and I fear we’ll have trouble holding the new line over time.” She further mused
“I wonder if this problem is, at least in part, due to how we make decisions. We don’t make them quickly enough, they aren’t clear enough, and we don’t have the most effective system of accountability.”
On one hand, traditional hospital culture is rightly grounded in the safety, hierarchy, and tradition of a do-no-harm world. But on the other hand, today’s economic, technological, and competitive environments require an approach to operations, revenue, and growth that has the aggressiveness of a Fortune 50 company. This should not be an either-or situation. Health systems can uphold a culture of safety while also fostering nontraditional values that will drive the organization assertively toward the future – all while committing to change.
Cartoon – Modern Decision Making
Cartoon – Operating Expense Reduction
Trinity Health to combine ministries, restructure leadership on West Coast
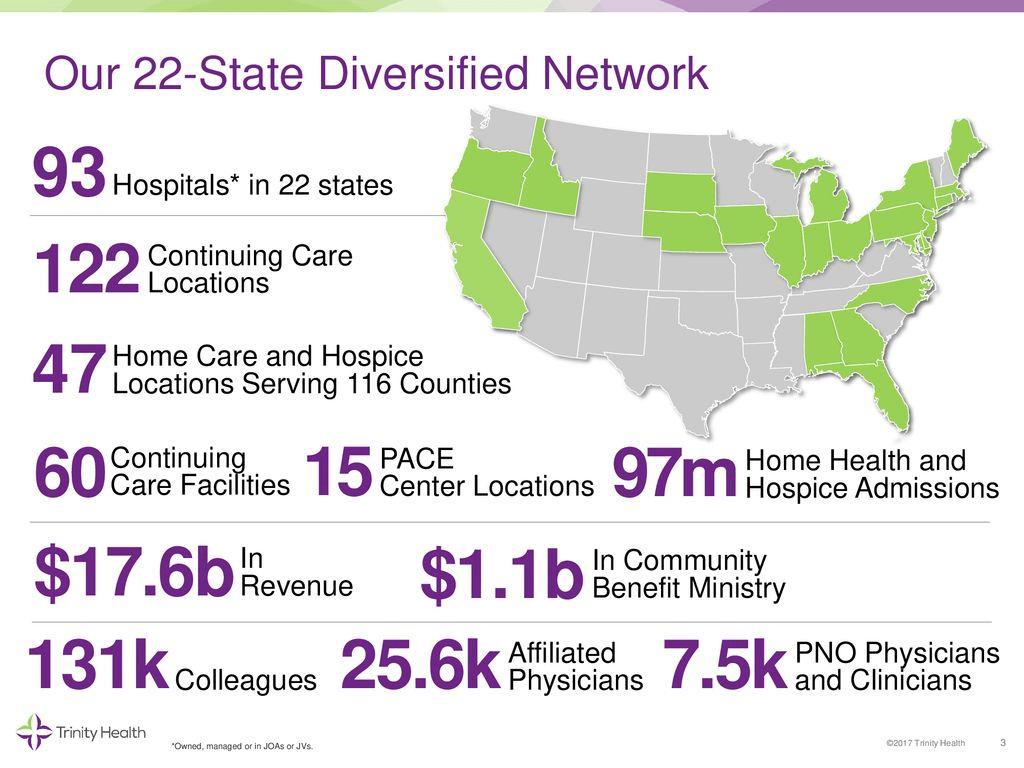
Livonia, Mich.-based Trinity Health is restructuring leadership on the West Coast as it combines Saint Agnes Medical Center in California and Saint Alphonsus Health System in Idaho and Oregon into one regional ministry, according to a statement shared with Becker’s May 4.
Trinity Health said the combination will allow these ministries “to streamline management and decision-making, reduce administrative costs and improve overall operating performance.”
The ministries will keep their names, and the boards of directors for each ministry will remain separate, the health system said. There will also be leadership changes.
Nancy Hollingsworth, MSN, RN, will retire as president and CEO of Fresno, Calif.-based Saint Agnes, effective May 26. Odette Bolano, BSN, president and CEO of Boise, Idaho-based Saint Alphonsus, will become president and CEO of the new regional entity. Additionally, David Spivey will join Saint Agnes as interim president and market leader.
This is a natural progression, as several services have already been consolidated between Saint Agnes and Saint Alphonsus, Trinity Health said.
The health system has also merged ministries in other regions, including Michigan, Indiana, Illinois, Iowa and New York.
Trinity Health has 123,000 employees in 26 states, according to its website.
Cartoon – Tracking Key Metrics
Cartoon – Talent Screening
Cartoon – Modern Decision Making
How can boards keep up with health system growth?
https://mailchi.mp/c6914989575d/the-weekly-gist-march-31-2023?e=d1e747d2d8

It feels like governance questions are coming to the fore in a lot of places these days, at least judging by several recent conversations we’ve had with health system CEOs. Probably not surprising, given the number of potential mergers and other partnerships under consideration.
As one CEO told us, growth by M&A raises particularly thorny issues for a not-for-profit system board. “Our governance structure grew out of a single hospital board, which was made up of community members and local physician leaders,” he told us. As the system acquired hospitals in adjacent markets, the combined board took on a representational character—each hospital had local stakeholders involved in governance. “Now we’re talking about merging with an out-of-state system, and our board suddenly seems way too parochial and unsophisticated. Everyone’s still asking what’s in it for their community.” That’s a frustration we hear frequently.
There are legitimate reasons why a tax-exempt community institution should have local representation on the board, advocating for local priorities and resources. But a larger, multi-state board must also oversee the entire portfolio of assets, and make trade-offs across markets, sometimes making decisions that favor one hospital over another.
The larger system board also has a greater need for sophistication, both on business and healthcare issues, as its members are often responsible for billions of dollars of assets. What frequently results from this tension is a nesting series of system and community boards, with varying degrees of accountability—a recipe for tangled, lengthy decision processes, and an enormous time-sink for senior system executives. We’re keeping our eye out for next-generation solutions to the governance question in healthcare—let us know what you’re seeing.







