
Cartoon – Modern Benefit Plans






The economic effects from the pandemic may place more pressure on investors to reevaluate the pay packages of CEOs in the future. But for the time being, “we are simply getting wealthier CEOs,” according to an annual report from the nonprofit shareholder advocacy group As You Sow.
For its report, As You Sow evaluated the most “overpaid” CEOs of S&P 500 companies. The nonprofit used data to compute what CEO pay would be assuming such pay is related to total shareholder return. In its methodology, a ranking of companies by excess CEO pay and by shareholder votes on CEO pay are weighted at 40 percent. The final ranking based on CEO-to-worker pay ratio is weighted at 20 percent. Find the full methodology here.
As You Sow notes some CEOs may no longer hold the positions listed below, as the rankings were calculated using data made available before June 30, 2020.
Here are 21 healthcare CEOs who made As You Sow’s list:
Larry Merlo (CVS Health)
Pay: $36.5 million
CEO-to-worker pay ratio: 790:1
Excess pay: $24.3 million
Alan Miller (King of Prussia, Pa.-based Universal Health Services)
Pay: $24.5 million
CEO-to-worker pay ratio: 629:1
Excess pay: $12.4 million
Michael Neidorff (Centene)
Pay: $26.4 million
CEO-to-worker pay ratio: 383:1
Excess pay: $13.3 million
Heather Bresch (Mylan)
Pay: $18.5 million
CEO-to-worker pay ratio: 427:1
Excess pay: $7.5 million
John Hammergren (McKesson)
Pay: $17.4 million
CEO-to-worker pay ratio: 458:1
Excess pay: $5.2 million
Samuel Hazen (Nashville, Tenn.-based HCA Healthcare)
Pay: $26.8 million
CEO-to-worker pay ratio: 478:1
Excess pay: $14.1 million
Stefano Pessina (Walgreens Boots Alliance)
Pay: $19.2 million
CEO-to-worker pay ratio: 562:1
Excess pay: $7.3 million
Ari Bousbib (IQVIA)
Pay: $22.1 million
CEO-to-worker pay ratio: 186:1
Excess pay: $8.7 million
Miles White (Abbott Laboratories)
Pay: $27.8 million
CEO-to-worker pay ratio: 329:1
Excess pay: $14.2 million
Javier Rodriguez (DaVita)
Pay: $16.9 million
CEO-to-worker pay ratio: 286:1
Excess pay: $4.3 million
Leonard Schleifer, MD, PhD (Regeneron Pharmaceuticals)
Pay: $21.5 million
CEO-to-worker pay ratio: 154:1
Excess pay: $8.6 million
Daniel O’Day (Gilead Sciences)
Pay: $29.1 million
CEO-to-worker pay ratio: 169:1
Excess pay: $16.9 million
David Cordani (Cigna)
Pay: $19.3 million
CEO-to-worker pay ratio: 306.7:1
Excess pay: $6.5 million
Michael Minogue (Abiomed)
Pay: $19.2 million
CEO-to-worker pay ratio: 166:1
Excess pay: $4.8 million
Joseph Hogan (Align Technology)
Pay: $18.3 million
CEO-to-worker pay ratio: 1,328:1
Excess pay: $3.5 million
Kenneth Frazier (Merck)
Pay: $27.6 million
CEO-to-worker pay ratio: 289:1
Excess pay: $14.5 million
Marc Casper (Thermo Fisher Scientific)
Pay: $19 million
CEO-to-worker pay ratio: 235:1
Excess pay: $5 million
Michel Vounatsos (Biogen)
Pay: $18.2 million
CEO-to-worker pay ratio: 114:1
Excess pay: $6 million
Michael Kaufmann (Cardinal Health)
Pay: $15.6 million
CEO-to-worker pay ratio: 272:1
Excess pay: $3.4 million
Vincent Forlenza (Becton, Dickinson and Co.)
Pay: $16 million
CEO-to-worker pay ratio: 379:1
Excess pay: $2.6 million
Omar Ishrak (Medtronic)
Pay: $17.8 million
CEO-to-worker pay ratio: 240:1
Excess pay: $4.8 million
Access the full list here.

Scientists at the Food and Drug Administration said Wednesday that the single-shot Covid-19 vaccine developed by Johnson & Johnson is effective and prevents hospitalizations from the disease.
Johnson & Johnson also revealed new, encouraging data showing the vaccine may do a better-than-expected job at protecting patients against new variants of the virus that causes disease. At the same time, FDA experts said the company’s study, results of which were originally made public in a Jan. 29 press release, includes insufficient information to draw conclusions on efficacy in people older than 75.
Documents from the FDA scientists, as well as separate documents from Johnson & Johnson, were released ahead of a Friday meeting of an FDA advisory panel in which outside experts will discuss and then vote on the risks and benefits of the new vaccine. The panel, known as the Vaccines and Related Biological Products Advisory Committee, makes recommendations to the FDA; the agency is not required to follow them, but it generally does.
The J&J vaccine is the first vaccine to show efficacy given as a single dose. It also does not need to be kept frozen when being shipped, as the vaccines developed by Moderna and the team of Pfizer and BioNTech do. Both of those advantages could be profound when it comes to vaccinating as many people as possible, a key step in slowing the spread of SARS-CoV-2.
Overall in the study, the vaccine reduced cases of Covid-19 that were rated as moderate to severe by 66.1% when considering cases occurring at least 28 days after vaccination. There were 193 cases that occurred at least 28 days after vaccination in the placebo group and 66 in the vaccine group. As of Feb. 5, there were seven Covid-19 related deaths in the placebo group and none in the vaccine group.
FDA researchers conducted a new analysis of how frequently volunteers in the study were hospitalized for Covid. When researchers counted cases 28 days after vaccination, there were zero hospitalizations in the vaccine arm and 16 in the placebo arm. For the full analysis set starting with the first dose, there were six hospitalizations for those who received the vaccine and 42 for those who did not.
Johnson & Johnson and the National Institutes of Health initially announced interim results of a 44,325 study testing the vaccine’s efficacy on Jan. 29. At the time, they said the 66% efficacy varied by geography. The vaccine was 72% protective in the U.S., compared to 58% in South Africa, where a new variant of SARS-CoV-2 is circulating.
In new documents, Johnson & Johnson said that in South Africa, the vaccine reduced severe or critical Covid-19 by 81.7% starting 28 days after vaccination, but that efficacy against more moderate disease was 64%. But the company said that the vaccine efficacy was not affected by the high prevalence of another variant in Brazil.
Unexpected side effects occurred at the same rate overall among volunteers who received vaccine and placebo — about 0.5%. However, some rare conditions appeared more common with the vaccine. Blood clot-related conditions occurred in 15 volunteers who received the vaccine and 10 who received placebo. Tinnitus, a ringing in the ears, occurred in six volunteers who received the vaccine and none who received placebo. The FDA said it will recommend monitoring for thromboembolic events after an EUA is granted.
Expected side effects that are related to the vaccine’s effect were common. Nearly half of volunteers reported injection site pain, 38.9% reported headache, 38.2% fatigue, and 33% reported muscle aches.
Johnson & Johnson also conducted an analysis in 2,650 volunteers looking at whether those who received the vaccine were less likely to test positive for the SARS-CoV-2 virus, which causes Covid-19, without having symptoms. There were 50 such cases in the placebo group compared to 18 among those who received the vaccine, a 65.5% reduction.
The United States has purchased 100 million doses of the vaccine, with an option to buy another 200 million doses. The agreement, announced last August, netted J&J over $1 billion in a contract with the Biomedical Advanced Research and Development Authority and the Department of Defense.
That said, the company currently has a limited number of doses to contribute to the effort to step up the country’s vaccine rollout. It will be April before J&J begins to have substantial amounts of vaccine to feed into the distribution pipeline, Moncef Slaoui, former co-chair of Operation Warp Speed, said earlier this year.
However, the company and the NIH said the vaccine was 85% effective at preventing severe disease, with no differences seen across the eight countries included in the study.
J&J is also conducting a trial in the United States of a two-dose vaccine, with the doses given eight weeks apart.The results from that 30,000 person trial are not expected until sometime in May.
The FDA documents represent the first close look at the data released Jan. 29, and are the result of a three-week effort by FDA scientists to independently evaluate the data generated in the trial. Friday’s panel will provide a deeper look at what those data actually mean.
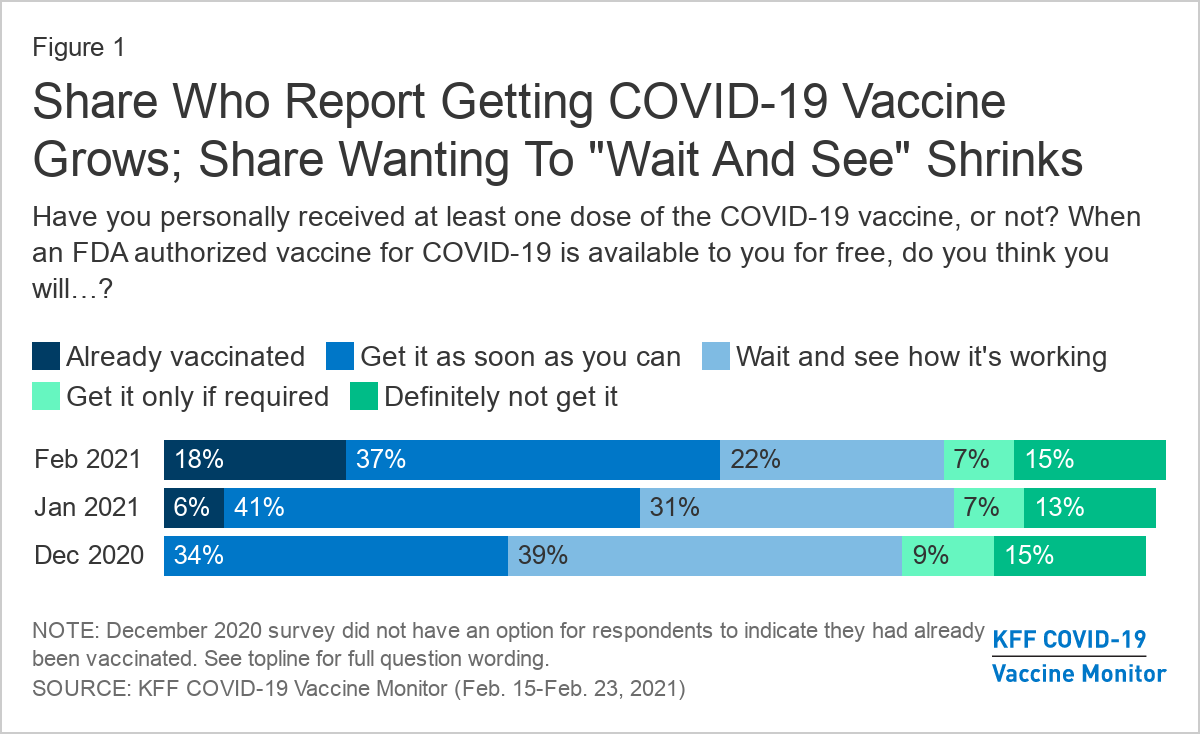
More than half of adults in the U.S. (55%) say they’ve already gotten one dose of Covid-19 vaccine or they’re eager to get one as soon as they can, an increase in acceptance from January (47%), a new poll reports. About 1 in 5 people are waiting to see how the vaccine rollout goes, but don’t rule out vaccination. Another 1 in 5 people are more reluctant: 7% would get vaccinated only if required by work, school, or some other activity, and 15% say no to vaccine under any circumstance. The increase in eagerness spans all demographic groups, but Black adults and young adults under age 30 were most likely to say they want to wait and see.
https://www.businessinsider.com/walmart-slowing-healthcare-clinics-strategy-2021-2
In 2018, Walmart‘s board of directors approved a bold plan to scale to 4,000 clinics by 2029.
The timeline laid out a net investment of $3 billion, not counting profits from the clinics, and a rollout strategy, according to a February 2019 presentation to the board obtained by Insider.
The vision was backed by former Walmart US CEO Greg Foran, the health team’s biggest champion who left Walmart in 2019. And it was dreamed up by Sean Slovenski, who Foran asked to come up with a big idea in healthcare as Walmart’s biggest competitors were pushing deeper into the space.
Now those leaders have been replaced by a team with a different philosophy, and the strategy is in flux at the same time Walmart is dealing with the pandemic and focusing on e-commerce, Insider has learned through conversations with eight former and current employees.
One coalition inside Walmart is happy with the change of pace —the retailer has 20 clinics currently, with at least 15 slotted for 2021 — because healthcare is hard, and the clinics are a work in progress.
Another coalition is frustrated by what they see as a stark departure from the initial goal to provide inexpensive care for people around the US quickly as possible.
Walmart didn’t comment on whether the rollout was slowing, but said it continued to “experiment” with Walmart Health centers and that the pandemic had reaffirmed its commitment to healthcare. It pointed to the launch of pharmacy curbside delivery, COVID-19 testing sites, and vaccine administration as evidence.

After filing a lawsuit in May to end its affiliation with Renton, Wash.-based Providence, Hoag Memorial Hospital in Newport Beach, Calif., is alleging it is now the target of retaliation, according to the Los Angeles Times.
Hoag Memorial said that Providence removed Hoag Memorial’s three facilities from its website of Southern California locations and terminated Hoag Memorial’s specialists from St. Joseph Heritage Healthcare, a network of medical providers for managed care plans in Southern California. Additionally, Hoag Memorial said that Providence informed Heritage members they would lose access to Hoag’s 13 urgent care centers by Dec. 31.
According to the report, Providence’s notice to patients that Hoag facilities and physicians would be dropped from its network all came in the fall of 2020, amid the COVID-19 pandemic.
“It was the most inappropriate, inexplicable and harsh thing to do to a lot of patients,” Hoag President and CEO Robert Braithwaite told the Los Angeles Times. “Finding a new physician or new specialist is particularly hard on seniors and any patient who has a chronic condition and has established a long-term relationship with an endocrinologist or rheumatologist or cancer doctor.”
Providence told the Los Angeles Times it disagrees that patients have been disadvantaged.
“We are committed to the well-being of our communities and to serving patients with high quality and compassionate care,” a Providence spokesperson told the Los Angeles Times.
Hoag Memorial has been affiliated with Providence, a Catholic health system, since 2016.
Hoag Memorial said the changes all came after the hospital sought to end its affiliation with Providence by filing a lawsuit. Hoag Memorial said in its lawsuit it is seeking to end the affiliation because Providence is undermining local decision-making and Catholic Church restrictions are expanding.
Providence has fought Hoag’s lawsuit to end the affiliation. The health system claims Hoag doesn’t have the right to unilaterally dissolve the affiliation, and its board members don’t have the authority to file the lawsuit. An Orange County Superior Court judge rejected Providence’s argument Feb. 1 and scheduled another court hearing for March.