
Category Archives: Vaccine Development
2023 Nobel Prize in Medicine: Karikó, Weissman awarded prize for mRNA research
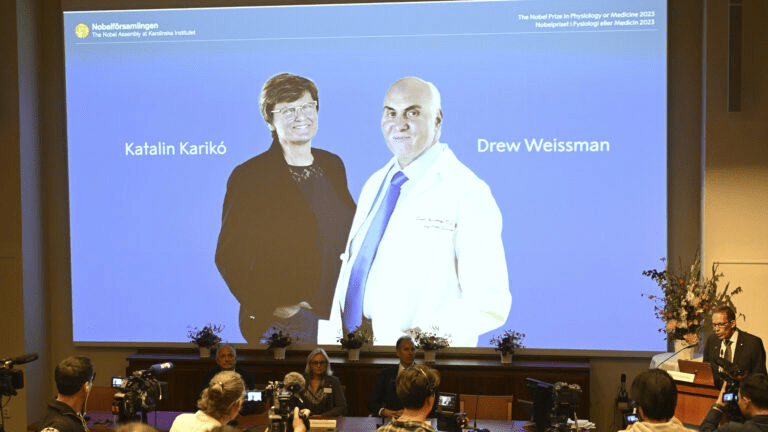
Two pioneers of mRNA research — the technology that helped the world tame the virus behind the Covid-19 pandemic — won the 2023 Nobel Prize in medicine or physiology on Monday.
Overcoming a lack of broader interest in their work and scientific challenges, Katalin Karikó and Drew Weissman made key discoveries about messenger RNA that enabled scientific teams to start developing the tool into therapies, immunizations, and — as the pandemic spread in 2020 — vaccines targeting the SARS-CoV-2 coronavirus. Moderna and the Pfizer-BioNTech partnership unveiled their mRNA-based Covid-19 shots in record time thanks to the foundational work of Karikó and Weissman, helping save millions of lives.
Karikó, a biochemist, and Weissman, an immunologist, performed their world-changing research on the interaction between mRNA and the immune system at the University of Pennsylvania, where Weissman, 64, remains a professor in vaccine research. Karikó, 68, who later went to work at BioNTech, is now a professor at Szeged University in her native Hungary, and is an adjunct professor at Penn’s Perelman School of Medicine.
The duo will receive 11 million Swedish kronor, or just over $1 million. Their names are added to a list of medicine or physiology Nobel winners that prior to this year included 213 men and 12 women.
The award was announced by Thomas Perlmann, secretary general of Nobel Assembly, in Stockholm. Perlmann said he had spoken to both laureates, describing them as grateful and surprised even though the pair has won numerous awards seen as precursors and had been tipped as likely Nobel recipients at some point.
Every year, the committee considers hundreds of nominations from former Nobel laureates, medical school deans, and prominent scientists from fields including microbiology, immunology, and oncology. Members try to identify a discovery that has altered scientists’ understanding of a subject. And according to the criteria laid out in Alfred Nobel’s will, that paradigm-shifting discovery also has to have benefited humankind.
The Nobel committee framed Karikó and Weissman’s work as a prime example of complementary expertise, with Karikó focused on RNA-based therapies and Weissman bringing a deep knowledge about immune responses to vaccines.
But it was not an easy road for the scientists. Karikó encountered rejection after rejection in the 1990s while applying for grants. She was even demoted while working at Penn, as she toiled away on the lower rungs of academia.
But the scientists persisted, and made a monumental discovery published in 2005 based on simply swapping out some of the components of mRNA.
With instructions from DNA, our cells make strands of mRNA that are then “read” to make proteins. The idea underlying an mRNA vaccine then is to take a piece of mRNA from a pathogen and slip it into our bodies. The mRNA will lead to the production of a protein from the virus, which our bodies learn to recognize and fight should we encounter it again in the form of the actual virus.
It’s an idea that goes back to the 1980s, as scientific advances allowed researchers to make mRNA easily in their labs. But there was a problem: The synthetic mRNA not only produced smaller amounts of protein than the natural version in our cells, it also elicited a potentially dangerous inflammatory immune response, and was often destroyed before it could reach target cells.
Karikó and Weissman’s breakthrough focused on how to overcome that problem. mRNA is made up of four nucleosides, or “letters”: A, U, G, and C. But the version our bodies make includes some nucleosides that are chemically modified — something the synthetic version didn’t, at least until Karikó and Weissman came along. They showed that subbing out some of the building blocks for modified versions allowed their strands of mRNA to sneak past the body’s immune defenses.
While the research did not gain wide attention at the time, it did catch the attention of scientists who would go on to found Moderna and BioNTech. And now, nearly 20 years later, billions of doses of mRNA vaccines have been administered.
For now, the only authorized mRNA products are the Covid-19 shots. But academic researchers and companies are exploring the technology as a potential therapeutic platform for an array of diseases and are using it to develop cancer vaccines as well as immunizations against other infectious diseases, from flu to mpox to HIV. An mRNA vaccine is highly adaptable compared to earlier methods, which makes it easier to alter the underlying recipe of the shot to keep up with viral evolution.
As she gained global fame, Karikó has been open about the barriers she encountered in her scientific career, which raised broader issues about the challenges women and immigrants can face in academia. But she’s said she always believed in the potential of her RNA research.
“I thought of going somewhere else, or doing something else,” Karikó told STAT in 2020, recalling the moment she was demoted. “I also thought maybe I’m not good enough, not smart enough. I tried to imagine: Everything is here, and I just have to do better experiments.”
American healthcare: The good, bad, ugly, future
https://www.linkedin.com/pulse/american-healthcare-good-bad-ugly-future-robert-pearl-m-d-/
Albert Einstein determined that time is relative. And when it comes to healthcare, five years can be both a long and a short amount of time.
In August 2018, I launched the Fixing Healthcare podcast. At the time, the medium felt like the perfect auditory companion to the books and articles I’d been writing. By bringing on world-renowned guests and engaging in difficult but meaningful discussions, I hoped the show would have a positive impact on American medicine. After five years and 100 episodes, now is an opportune time to look back and examine how healthcare has improved and in what ways American medicine has become more problematic.
Here’s a look at the good, the bad and the ugly since episode one of Fixing Healthcare:
The Good
Drug breakthroughs and government actions headline medicine’s biggest wins over the past five years.
Vaccines
Arguably the most massive (and controversial) healthcare triumph over the past five years was the introduction of vaccines, which proved successful beyond any reasonable expectation.
At first, health experts expressed doubts that Pfizer, Moderna and others could create a safe and effective Covid-19 vaccine with messenger RNA (mRNA) technology. After all, no one had succeeded in more than two decades of trying.
Thanks in part to Operation Warp Speed, the government-funded springboard for research, our nation produced multiple vaccines within less than a year. Previously, the quickest vaccine took four years to develop (mumps). All others required a minimum of five years.
The vaccines were pivotal in ending the coronavirus pandemic, and their success has opened the door to other life-saving drugs, including those that might prevent or fight cancer. And, of course, our world is now better prepared for when the next viral pandemic strikes.
Weight-Loss Drugs
Originally designed to help patients manage Type 2 diabetes, drugs like Ozempic have been helping people reverse obesity—a condition closely correlated with diabetes, heart disease and cancer.
For decades, America’s $150 billion a year diet industry has failed to curb the nation’s continued weight gain. So too have calls for increased exercise and proper nutrition, including restrictions on sugary sodas and fast foods.
In contrast, these GLP-1 medications are highly effective. They help overweight and obese people lose 15 to 25 pounds on average with side effects that are manageable for nearly all users.
The biggest stumbling block to their widespread use is the drug’s exorbitant price (upwards of $16,000 for a year’s supply).
Drug-Pricing Laws
With the Inflation Reduction Act of 2022, Congress took meaningful action to lower drug prices, a move the CBO estimates would reduce the federal deficit by $237 billion over 10 years.
It’s a good start. Americans today pay twice as much for the same medications as people in Europe largely because of Congressional legislation passed in 2003.
That law, the Medicare Prescription Drug Price Negotiation Act, made it illegal for Health and Human Services (HHS) to negotiate drug prices with manufacturers—even for the individuals publicly insured through Medicare and Medicaid.
Now, under provisions of the new Inflation Reduction Act, the government will be able to negotiate the prices of 10 widely prescribed medications based on how much Medicare’s Part D program spends. The lineup is expected to include prescription treatments for arthritis, cancer, asthma and cardiovascular disease. Unfortunately, the program won’t take effect until 2026. And as of now, several legal challenges from both drug manufacturers and the U.S. Chamber of Commerce are pending.
The Bad
Spiking costs, ongoing racial inequalities and millions of Americans without health insurance make up three disappointing healthcare failures of the past five years.
Cost And Quality
The U.S. spends nearly twice as much on healthcare per citizen as other countries, yet our nation lags 10 of the wealthiest countries in medical performance and clinical outcomes. As a result, Americans die younger and experience more complications from chronic diseases than people in peer nations.
As prices climb ever-higher, at least half of Americans can’t afford to pay their out-of-pocket medical bills, which remain the leading cause of U.S. bankruptcy. And with rising insurance premiums alongside growing out-of-pocket expenses, more people are delaying their medical care and rationing their medications, including life-essential drugs like insulin. This creates a vicious cycle that will likely prolong today’s healthcare problems well into the future.
Health Disparities
Inequalities in American medicine persist along racial lines—despite action-oriented words from health officials that date back decades.
Today, patients in minority populations receive unequal and inequitable medical treatment when compared to white patients. That’s true even when adjusting for differences in geography, insurance status and socioeconomics.
Racism in medical care has been well-documented throughout history. But the early days of the Covid-19 pandemic provided several recent and deadly examples. From testing to treatment, Black and Latino patients received both poorer quality and less medical care, doubling and even tripling their chances of dying from the disease.
The problems can be observed across the medical spectrum. Studies show Black women are still less likely to be offered breast reconstruction after mastectomy than white women. Research also finds that Black patients are 40% less likely to receive pain medication after surgery. Although technology could have helped to mitigate health disparities, our nation’s unwillingness to acknowledge the severity of the problem has made the problem worse.
Uninsurance
Although there are now more than 90 million Americans enrolled in Medicaid, there are still 30 million people without any health insurance. This disturbing reality comes a full decade after the passage of the Affordable Care Act.
On Capitol Hill, there is no plan in place to reduce the number of uninsured.
Moreover, many states are looking to significantly rollback their Medicaid enrollment in the post-Covid era. Kaiser Family Foundation estimates that between 8 million and 24 million people will lose Medicaid coverage during the unwinding of the continuous enrollment provisions implemented during the pandemic. Without coverage, people have a harder time obtaining the preventive services they need and, as a result, they suffer more chronic diseases and die younger.
The Ugly
An overall decrease in longevity, along with higher maternal mortality and a worsening mental-health crisis, comprise the greatest failures of U.S. healthcare over the past five years.
Life Expectancy
Despite radical advances in medical science over the past five years, American life expectancy is back to where it was at the turn of the 20th century, according to CDC data.
Alongside environmental and social factors are a number of medical causes for the nation’s dip in longevity. Research demonstrated that many of the 1 million-plus Covid-19 deaths were preventable. So, too, was the nation’s rise in opioid deaths and teen suicides.
Regardless of exact causation, Americans are living two years less on average than when we started the Fixing Healthcare podcast five years ago.
Maternal Mortality
Compared to peer nations, the United States is the only country with a growing rate of mothers dying from childbirth. The U.S. experiences 17.4 maternal deaths per 100,000 live births. In contrast, Norway is at 1.8 and the Netherlands at 3.0.
The risk of dying during delivery or in the post-partum period is dramatically higher for Black women in the United States. Even when controlling for economic factors, Black mothers still suffer twice as many deaths from childbirth as white women.
And with growing restrictions on a woman’s right to choose, the maternal mortality rate will likely continue to rise in the United States going forward.
Mental Health
Finally, the mental health of our country is in decline with rates of anxiety, depression and suicide on the rise.
These problems were bad prior to Covid-19, but years of isolation and social distancing only aggravated the problem. Suicide is now a leading cause of death for teenagers. Now, more than 1 in every 1,000 youths take their own lives each year. The newest data show that suicides across the U.S. have reached an all-time high and now exceed homicides.
Even with the expanded use of telemedicine, mental health in our nation is likely to become worse as Americans struggle to access and afford the services they require.
The Future
In looking at the three lists, I’m reminded of a baseball slugger who can occasionally hit awe-inspiring home runs but strikes out most of the time. The crowd may love the big hitter and celebrate the long ball, but in both baseball and healthcare, failing at the basics consistently results in more losses than wins.
Over the past five years, American medicine has produced a losing record. New drugs and surgical breakthroughs have made headlines, but the deeper, more systemic failures of American healthcare have rarely penetrated the news cycle.
If our nation wants to make the next five years better and healthier than the last five, elected officials and healthcare leaders will need to make major improvements. The steps required to do so will be the focus of my next article.
The end of the pandemic “is in sight”

Some good news: The world had its lowest COVID death toll last week since March 2020, the World Health Organization said.
- The end of the pandemic “is in sight,” said WHO Director-General Tedros Adhanom Ghebreyesus.
- But “we are not there yet.”
Zoom out: Last summer’s Delta variant demolished the first sense of relief after vaccines.
- “If we don’t take this opportunity now, Tedros said while calling for more vaccinations and testing, “we run the risk of more variants, more deaths, more disruption and more uncertainty.”
The bottom line: The next surge could come by surprise.
- Johns Hopkins University is scaling back its COVID metrics due to a slowdown in local data reporting, the university confirmed to Axios.
FDA clears updated COVID-19 vaccines ahead of fall booster campaign

The Food and Drug Administration (FDA) on Wednesday authorized updated COVID-19 booster shots specifically targeting a subvariant of omicron.
The move comes ahead of a fall campaign to give Americans booster shots, which is expected to launch in the coming days.
The move marks the first time the vaccines have been updated since the first shots were cleared at the end of 2020, and the updated shots are designed to catch up to evolutions in the virus.
The shots from Pfizer and Moderna target the omicron subvariants BA.4 and BA.5, as well as the original virus.
The shots can begin going into arms once the final step in the process, a Centers for Disease Control and Prevention committee, clears them, which is expected to occur on Thursday.
A major question, though, is how many people will actually want the new shots, given that uptake for the existing booster shots has lagged.
Only about half of people who got the first two shots received the initial booster dose.
“The COVID-19 vaccines, including boosters, continue to save countless lives and prevent the most serious outcomes (hospitalization and death) of COVID-19,” said FDA Commissioner Robert Califf. “As we head into fall and begin to spend more time indoors, we strongly encourage anyone who is eligible to consider receiving a booster dose with a bivalent COVID-19 vaccine to provide better protection against currently circulating variants.”
Seeking to keep up with the ever-evolving virus, the FDA did not wait for the time-consuming process of going through full clinical trials on this tweaked vaccine. But it noted that it is highly confident that the vaccines are safe and effective. The agency pointed to the millions of doses of the original vaccines that have been given, as well as data from another version of the updated vaccine, along with preliminary data on this one.
Peter Marks, a top FDA vaccine official, compared the process to the annual updates to the flu vaccine that seek to adapt to the changes in that virus.
“The public can be assured that a great deal of care has been taken by the FDA to ensure that these bivalent COVID-19 vaccines meet our rigorous safety, effectiveness and manufacturing quality standards for emergency use authorization,” Marks said.
The updated Moderna vaccine is cleared for people 18 and older, and the Pfizer vaccine for people 12 and older.
For both, people are eligible for the booster shot of the updated vaccine if it has been at least two months since their last shot.
BA.5 spurs new calls to fund next-generation COVID-19 vaccines

The rise of the BA.5 variant is spurring new calls for funding for an Operation Warp Speed 2.0 to accelerate development of next-generation COVID-19 vaccines that can better target new variants.
The BA.5 subvariant of omicron that now makes up the majority of U.S. COVID-19 cases is sparking concern because it has a greater ability to evade the protection of current vaccines than past strains of the virus did.
Pfizer and Moderna are working on updated vaccines that target BA.5 that could be ready this fall, but experts say that by the time they are ready, a new variant very well could have taken hold.
As alternatives to vaccine makers chasing each variant, experts point to research on “pan-coronavirus” vaccines that are “variant-proof,” targeting multiple variants, as well as nasal vaccines that could drastically cut down on transmission of the virus.
There is ongoing research on these next-generation vaccines, but unlike in 2020, when the federal government’s Operation Warp Speed helped speed the development of the original vaccine, there is less funding and assistance this time around.
COVID-19 funding that could help develop and manufacture new vaccines more quickly has been stalled in Congress for months.
“There’s no Operation Warp Speed,” said Eric Topol, professor of molecular medicine at Scripps Research. “So it’s moving very slowly. But at least it’s moving.”
Leana Wen, a public health professor at George Washington University, wrote in a Washington Post op-ed this week that the U.S. needs “urgent investment” in next-generation vaccines and “we need an ‘Operation Warp Speed Part 2.’”
Pfizer and Moderna are working on updated vaccines that target BA.5 that could be ready this fall, but experts say that by the time they are ready, a new variant very well could have taken hold.
As alternatives to vaccine makers chasing each variant, experts point to research on “pan-coronavirus” vaccines that are “variant-proof,” targeting multiple variants, as well as nasal vaccines that could drastically cut down on transmission of the virus.
There is ongoing research on these next-generation vaccines, but unlike in 2020, when the federal government’s Operation Warp Speed helped speed the development of the original vaccine, there is less funding and assistance this time around.
COVID-19 funding that could help develop and manufacture new vaccines more quickly has been stalled in Congress for months.
“There’s no Operation Warp Speed,” said Eric Topol, professor of molecular medicine at Scripps Research. “So it’s moving very slowly. But at least it’s moving.”
Leana Wen, a public health professor at George Washington University, wrote in a Washington Post op-ed this week that the U.S. needs “urgent investment” in next-generation vaccines and “we need an ‘Operation Warp Speed Part 2.’”
Administration health officials pointed to funding when asked about next-generation vaccines at a press briefing on Tuesday.
“We need resources to continue that effort and to accelerate that effort,” said Anthony Fauci, the government’s top infectious disease expert. “So although we’re doing a lot and the field looks promising, in order to continue it, we really do need to have a continual flow of resources to do that.”
But COVID-19 funding has been stuck in Congress for months. Republicans have long said they do not see any urgency in approving the money. Democrats, while generally calling for the funding, have been caught up in their own internal divisions, like when a group of House Democrats objected to a way to pay for the new funding in March.
“Of course more funding would accelerate some parts of the development,” Karin Bok, acting deputy director of the National Institutes of Health’s (NIH) Vaccine Research Center, said in an interview.
She also cautioned that development of next-generation vaccines like nasal vaccines would take longer than the original vaccines, because less groundwork has been laid over the preceding years.
Experts stress that even for BA.5, the current vaccines still provide important protection against severe disease and hospitalization, and are urging people to get their booster shots now. But there is potential for further improvement in the vaccines as well.
Aside from funding, another obstacle is obtaining copies of the existing COVID-19 vaccines for use in research, said Pamela Bjorkman, a California Institute of Technology professor working on a next-generation vaccine.
“I would say we’ve wasted at least six months,” with various procedural hurdles on that front, she said. “It’s just ridiculous.”
For example, she said at one point when her team was able to get access to the AstraZeneca COVID-19 vaccine, it then took two or three months to get an import permit to send it from the United Kingdom.
“This is a hot topic,” Bok, of the NIH, said of access to existing vaccine doses for researchers. “The government is working very hard on an agreement with the companies to provide it to us and to all the investigators…that are funded by NIH.”
Asked about providing vaccine doses for researchers and any talks with the administration on that front, a Moderna spokesperson said: “We do provide vaccine in certain investigator-initiated studies where physicians and scientists propose research they have designed and want to conduct with our support,” pointing to a South African study as an example.
More broadly, the White House says it is working on accelerating next-generation vaccine research and will have more announcements soon.
“Let me be very clear: We clearly need a true next-generation vaccine,” White House COVID-19 response coordinator Ashish Jha told reporters on Tuesday.
“You’ll hear more from us in the days and weeks ahead,” he added. “This is something that we have been working quite assiduously on.”
COVID vaccine strategy still murky after FDA experts meet
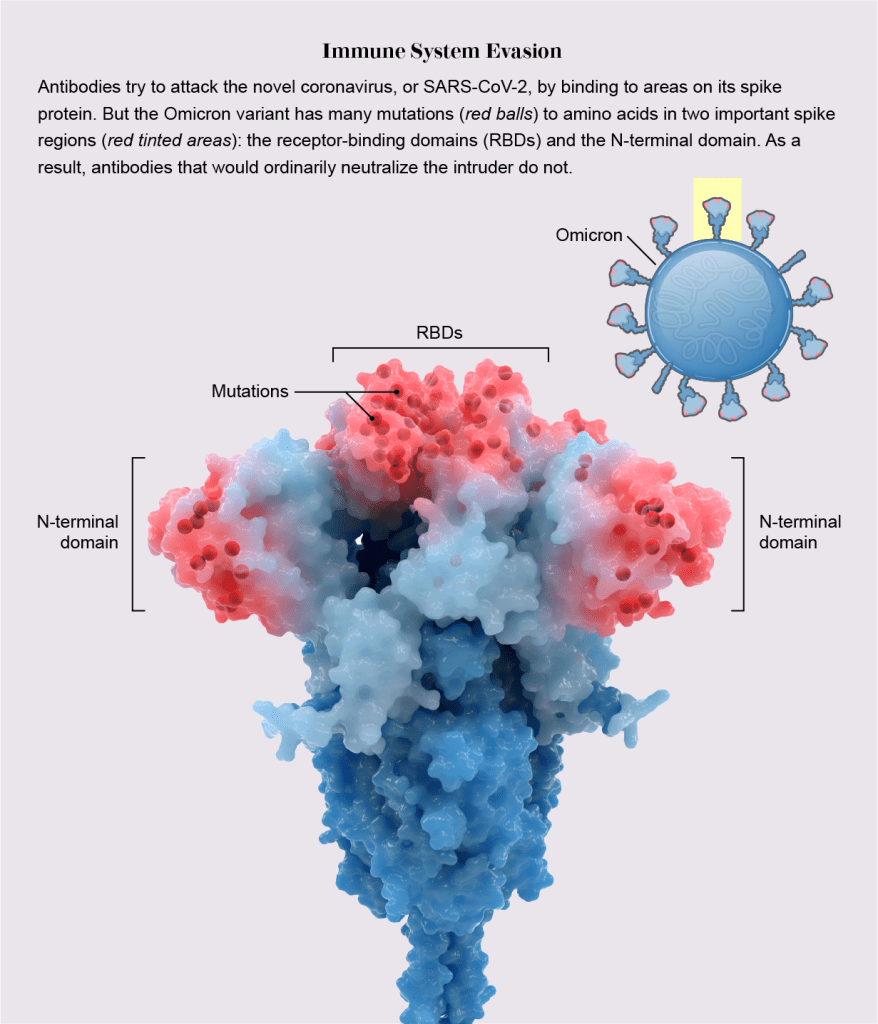
The COVID-19 vaccine strategy for the fall remains beset with unanswered questions after an FDA expert panel on Tuesday spent hours debating how and whether to update the shots.
Why it matters: Time is running short to develop a game plan with existing vaccines losing effectiveness against new variants and more than half of Americans still without a booster dose.
Driving the news: The Vaccines and Related Biological Products Advisory Committee voted 19-2 to recommend an Omicron-specific update to COVID-19 booster vaccines expected to be rolled out within the next few months.
But key questions were left unanswered:
- The panel didn’t formally decide whether to update shots with the prevalent Omicron strain in circulation, currently the fast-spreading BA.4 and BA.5 subvariants, or the BA.1 lineage that emerged late last year, as the World Health Organization recommended.
- The consensus appeared to be for a bivalent, or combination, booster combining the original COVID-19 strain that emerged in late 2019 with BA.4 and BA.5.
- The FDA will continue to evaluate what to do about the primary series of vaccines for the fall.
- Experts were split on whether there was enough data to recommend the updated shots for kids, or whether more studies are needed on dosage and possible side effects.
- There also were concerns about what effect an updated vaccine would have on developing nations’ willingness to use older COVID shots to inoculate their populations.
- And above all, it’s unclear whether all the questions about who gets which shot when will add to public confusion and apathy that’s dogged the vaccination effort in recent months.
What they’re saying: “None of us has a crystal ball and we’re trying to use every last ounce of what we can from predictive modeling and data that’s emerging to try to get ahead of a virus that’s very crafty,” said top FDA vaccine regulator Peter Marks.
- “Unfortunately, looking in the past doesn’t help us a great deal to look in the future for [a] virus that has baffled a lot of us and made predictions almost irrelevant,” said acting panel chairman Arnold Monto, a University of Michigan epidemiologist.
The timetable: Pfizer-BioNTech said an updated mRNA vaccine could be ready in October if regulatory uncertainties are ironed out. Moderna said it could have large amounts ready in late October or early November. Novavax is still awaiting emergency use authorization for its protein-based shot, but said it could have an updated vaccine by the fourth quarter.
Failing to fund the U.S. covid response bodes trouble for the entire world

Atul Gawande leads global health and is co-chair of the Covid-19 Task Force at the U.S. Agency for International Development.
Nearly a year ago, President Biden announced that the United States would be the “arsenal of vaccines for the world,” just as America served as an arsenal for democracies during World War II. With the president’s leadership and the consistent bipartisan support of Congress, the United States has delivered more than half a billion coronavirus vaccines to 114 lower-income countries free of charge, a historic accomplishment. This example spurred contributions from other wealthy nations and contributed to vaccination of almost 60 percent of the world.
But the global battle against covid-19 is not done. Instead, the challenge has changed. The lowest-income countries, where vaccinations have reached less than 15 percent of people, are now declining free vaccine supply because they don’t have the capacity to get shots in arms fast enough.
We must therefore not just provide an arsenal; to protect our allies against future variants, we must also provide the support they need to ramp up their vaccination campaigns. That effort requires money, and despite generously funding our covid-19 response up to this point, Congress is now failing to provide the resources we need.
I am writing to say: This bodes serious trouble for the world.
Despite a period of relative calm here at home, we’re again seeing cases and hospitalizations spike in Europe and Asia, even in places with higher levels of vaccination than the United States. These surges are due to the more-transmissible BA.2 subvariant of the already highly infectious omicron strain. Without additional funding, we risk not having the tools we need — vaccines, treatments, tests, masks and more — to manage future surges at home. And no less troubling, if we don’t close the vaccine gap between richer and poorer countries, we will give the virus more chances to mutate into a new variant.
Since the virus first emerged, the package of tools we’ve developed to fight it has proved resilient against all coronavirus variants. But there’s no guarantee that will remain true. A new variant that evades our defenses might once again fuel new surges of severe illness and batter the global economy. Helping all countries protect their populations by supercharging vaccination campaigns is our best hope to prevent future strains from emerging and ending this pandemic once and for all.
Turning vaccines into actual vaccinations has been difficult even in wealthy countries, where capable health systems, state-of-the-art cold chains and public awareness campaigns mean that anyone who wants a vaccine can get one. In countries without strong health infrastructure — without enough freezers and refrigerated trucks to keep vaccines from spoiling or enough health-care workers to reach rural populations living miles from the nearest health facility — it’s much tougher. We’ve also seen the same vaccine myths and disinformation that swirl through our media ecosystem spread just as rapidly through social media and hurt public trust abroad.
But we’ve also learned how to successfully tackle these challenges. In December, the Biden administration launched an initiative called Global VAX to help low-income countries train health workers, strengthen health infrastructure and raise vaccine access and awareness. While vaccine coverage in those countries remains far below the global average, the rapid progress we’ve supported in places such as Ivory Coast, Uganda and Zambia show what is possible when governments that are committed to fighting covid-19 have the global support they need.
Without more funding, we would have to halt our plans to expand the Global VAX initiative. The United States would have to turn its back on countries that need urgent help to boost their vaccination rates. And many countries that finally have the vaccines they need to protect their populations would risk seeing them spoil on the tarmac.
We can’t let this happen. It not only endangers people abroad but also risks the health and prosperity of all Americans. The virus is not waiting on Congress to negotiate; it is infecting people and mutating as we speak.
Over the past two years, both parties in Congress have repeatedly stepped up to fight covid-19 in an inspiring show of bipartisan unity. Now, we need our leaders to come together once more. With an effective strategy in place and the tools to transform covid-19 from a killer pandemic to a manageable respiratory disease, the United States has the expertise and capabilities the world needs to win the fight against this virus. We need Congress to let us take the fight to the front lines.
The Next Big COVID Variant Could Be a Triple Whammy Nightmare
https://www.yahoo.com/news/next-big-covid-variant-could-100250868.html
Even as daily new COVID cases set all-time records and hospitals fill up, epidemiologists have arrived at a perhaps surprising consensus. Yes, the latest Omicron variant of the novel coronavirus is bad. But it could have been a lot worse.
Even as cases have surged, deaths haven’t—at least not to the same degree. Omicron is highly transmissible but generally not as severe as some older variants—“lineages” is the scientific term.
We got lucky. But that luck might not hold. Many of the same epidemiologists who have breathed a sigh of relief over Omicron’s relatively low death rate are anticipating that the next lineage might be much worse.
The New Version of the Omicron Variant Is a Sneaky Little Bastard
Fretting over a possible future lineage that combines Omicron’s extreme transmissibility with the severity of, say, the previous Delta lineage, experts are beginning to embrace a new public health strategy that’s getting an early test run in Israel: a four-shot regimen of messenger-RNA vaccine.
“I think this will be the strategy going forward,” Edwin Michael, an epidemiologist at the Center for Global Health Infectious Disease Research at the University of South Florida, told The Daily Beast.
Omicron raised alarms in health agencies all over the world in late November after officials in South Africa reported the first cases. Compared to older lineages, Omicron features around 50 key mutations, some 30 of which are on the spike protein that helps the virus to grab onto our cells.
Some of the mutations are associated with a virus’s ability to dodge antibodies and thus partially evade vaccines. Others are associated with higher transmissibility. The lineage’s genetic makeup pointed to a huge spike in infections in the unvaccinated as well as an increase in milder “breakthrough” infections in the vaccinated.
That’s exactly what happened. Health officials registered more than 10 million new COVID cases the first week of January. That’s nearly double the previous worst week for new infections, back in May. Around 3 million of those infections were in the United States, where Omicron coincided with the Thanksgiving, Christmas, and New Year holidays and associated traveling and family gatherings.
But mercifully, deaths haven’t increased as much as cases have. Worldwide, there were 43,000 COVID deaths the first week of January—fewer than 10,000 of them in the U.S. While deaths tend to lag infections by a couple weeks, Omicron has been dominant long enough that it’s increasingly evident there’s been what statisticians call a “decoupling” of cases and fatalities.
“We can say we dodged a bullet in that Omicron does not appear to cause as serious of a disease,” Stephanie James, the head of a COVID testing lab at Regis University in Colorado, told The Daily Beast. She stressed that data is still being gathered, so we can’t be certain yet that the apparent decoupling is real.
Assuming the decoupling is happening, experts attribute it to two factors. First, Omicron tends to infect the throat without necessarily descending to the lungs, where the potential for lasting or fatal damage is much, much higher. Second, by now, countries have administered nearly 9.3 billion doses of vaccine—enough for a majority of the world’s population to have received at least one dose.
Omicron Shows the Unvaccinated Will Never Be Safe
In the United States, 73 percent of people have gotten at least one dose. Sixty-two percent have gotten two doses of the best mRNA vaccines. A third have received a booster dose.
Yes, Omicron has some ability to evade antibodies, meaning the vaccines are somewhat less effective against this lineage than they are against Delta and other older lineages. But even when a vaccine doesn’t prevent an infection, it usually greatly reduces its severity.
For many vaccinated people who’ve caught Omicron, the resulting COVID infection is mild. “A common cold or some sniffles in a fully vaxxed and boosted healthy individual,” is how Eric Bortz, a University of Alaska-Anchorage virologist and public health expert, described it to The Daily Beast.
All that is to say, Omicron could have been a lot worse. Viruses evolve to survive. That can mean greater transmissibility, antibody-evasion or more serious infection. Omicron mutated for the former two. There’s a chance some future Sigma or Upsilon lineage could do all three.
When it comes to viral mutations, “extreme events can occur at a non-negligible rate, or probability, and can lead to large consequences,” Michael said. Imagine a lineage that’s as transmissible as Omicron but also attacks the lungs like Delta tends to do. Now imagine that this hypothetical lineage is even more adept than Omicron at evading the vaccines.
2022’s Hottest New Illness: Flurona
That would be the nightmare lineage. And it’s entirely conceivable it’s in our future. There are enough vaccine holdouts, such as the roughly 50 million Americans who say they’ll never get jabbed, that the SARS-CoV-2 pathogen should have ample opportunities for mutation.
“As long as we have unvaccinated people in this country—and across the globe—there is the potential for new and possibly more concerning viral variants to arise,” Aimee Bernard, a University of Colorado immunologist, told The Daily Beast.
Worse, this ongoing viral evolution is happening against a backdrop of waning immunity. Antibodies, whether vaccine-induced or naturally occurring from past infection, fade over time. It’s not for no reason that health agencies in many countries urge booster doses just three months after initial vaccination. The U.S. Centers for Disease Control and Prevention is an outlier, and recommends people get boosted after five months.
A lineage much worse than Omicron could evolve at the same time that antibodies wane in billions of people all over the world. That’s why many experts believe the COVID vaccines will end up being annual or even semi-annual jabs. You’ll need a fourth jab, a fifth jab, a sixth jab, et cetera, forever.
Israel, a world leader in global health, is already turning that expectation into policy. Citing multiple studies that showed a big boost in antibodies with an additional dose of mRNA and no safety concerns, the country’s health ministry this week began offering a fourth dose to anyone over the age of 60, who tend to be more vulnerable to COVID than younger people.
That should be the standard everywhere, Ali Mokdad, a professor of health metrics sciences at the University of Washington Institute for Health, told The Daily Beast. “Scientifically, they’re right,” he said of the Israeli health officials.
If there’s a downside, it’s that there are still a few poorer countries—in Africa, mostly—where many people still struggle to get access to any vaccine, let alone boosters and fourth doses. If and when other richer countries follow Israel’s lead and begin offering additional jabs, there’s some risk of even greater inequity in global vaccine distribution.
“The downside is for the rest of the world,” Mokdad said. “I’m waiting to get my first dose and you guys are getting a fourth?”
The solution isn’t to deprive people of the doses they need to maintain their protection against future—and potentially more dangerous—lineages. The solution, for vaccine-producing countries, is to further boost production and double down on efforts to push vaccines out to the least privileged communities.
A sense of urgency is key. For all its rapid spread, Omicron has actually gone fairly easy on us. Sigma or Upsilon might not.
PFIZER AND BIONTECH PROVIDE UPDATE ON OMICRON VARIANT

- Preliminary laboratory studies demonstrate that three doses of the Pfizer-BioNTech COVID-19 Vaccine neutralize the Omicron variant (B.1.1.529 lineage) while two doses show significantly reduced neutralization titers
- Data indicate that a third dose of BNT162b2 increases the neutralizing antibody titers by 25-fold compared to two doses against the Omicron variant; titers after the booster dose are comparable to titers observed after two doses against the wild-type virus which are associated with high levels of protection
- As 80% of epitopes in the spike protein recognized by CD8+ T cells are not affected by the mutations in the Omicron variant, two doses may still induce protection against severe disease
- The companies continue to advance the development of a variant-specific vaccine for Omicron and expect to have it available by March in the event that an adaption is needed to further increase the level and duration of protection – with no change expected to the companies’ four billion dose capacity for 2022
NEW YORK & MAINZ, Germany–(BUSINESS WIRE)– Pfizer Inc. (NYSE: PFE) and BioNTech SE (Nasdaq: BNTX) today announced results from an initial laboratory study demonstrating that serum antibodies induced by the Pfizer-BioNTech COVID-19 Vaccine (BNT162b2) neutralize the SARS-CoV-2 Omicron variant after three doses. Sera obtained from vaccinees one month after receiving the booster vaccination (third dose of BNT162b2 vaccine) neutralized the Omicron variant to levels that are comparable to thoseobserved for the wild-type SARS-CoV-2 spike protein after two doses.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20211208005542/en/
Sera from individuals who received two doses of the current COVID-19 vaccine did exhibit, on average, more than a 25-fold reduction in neutralization titers against the Omicron variant compared to wild-type, indicating that two doses of BNT162b2 may not be sufficient to protect against infection with the Omicron variant. However, as the vast majority of epitopes targeted by vaccine-induced T cells are not affected by the mutations in Omicron, the companies believe that vaccinated individuals may still be protected against severe forms of the disease and are closely monitoring real world effectiveness against Omicron, globally.
A more robust protection may be achieved by a third dose as data from additional studies of the companies indicate that a booster with the current COVID-19 vaccine from Pfizer and BioNTech increases the antibody titers by 25-fold. According to the companies’ preliminary data, a third dose provides a similar level of neutralizing antibodies to Omicron as is observed after two doses against wild-type and other variants that emerged before Omicron. These antibody levels are associated with high efficacy against both the wild-type virus and these variants. A third dose also strongly increases CD8+ T cell levels against multiple spike protein epitopes which are considered to correlate with the protection against severe disease. Compared to the wild-type virus, the vast majority of these epitopes remain unchanged in the Omicron spike variant.
“Although two doses of the vaccine may still offer protection against severe disease caused by the Omicron strain, it’s clear from these preliminary data that protection is improved with a third dose of our vaccine,” said Albert Bourla, Chairman and Chief Executive Officer, Pfizer. “Ensuring as many people as possible are fully vaccinated with the first two dose series and a booster remains the best course of action to prevent the spread of COVID-19.”
“Our preliminary, first dataset indicate that a third dose could still offer a sufficient level of protection from disease of any severity caused by the Omicron variant,” said Ugur Sahin, M.D., CEO and Co-Founder of BioNTech. “Broad vaccination and booster campaigns around the world could help us to better protect people everywhere and to get through the winter season. We continue to work on an adapted vaccine which, we believe, will help to induce a high level of protection against Omicron-induced COVID-19 disease as well as a prolonged protection compared to the current vaccine.”
While these results are preliminary, the companies will continue to collect more laboratory data and evaluate real-world effectiveness to assess and confirm protection against Omicron and inform the most effective path forward. On November 25, the companies started to develop an Omicron-specific COVID-19 vaccine. The development will continue as planned in the event that a vaccine adaption is needed to increase the level and duration of protection against Omicron. First batches of the Omicron-based vaccine can be produced and are planned to be ready for deliveries within 100 days, pending regulatory approval. Pfizer and BioNTech have tested other variant-specific vaccines as well, which have produced very strong neutralization titers and a tolerable safety profile. Based on this experience the companies have high confidence that if needed they can deliver an Omicron-based vaccine in March 2022. The companies have also previously initiated clinical trials with variant-specific vaccines (Alpha, Beta, Delta & Alpha/Delta Mix) and data from these studies will be submitted to regulatory agencies around the world to help accelerate the process of adapting the vaccine and gaining regulatory authorization or approval of an Omicron-specific vaccine, if needed. The companies have previously announced that they expect to produce four billion doses of BNT162b2 in 2022, and this capacity is not expected to change if an adapted vaccine is required.
About the Pfizer-BioNTech Laboratory Studies
To evaluate the effectiveness of BNT162b2 against the Omicron variant, Pfizer and BioNTech immediately tested a panel of human immune sera obtained from the blood of individuals that received two or three 30-µg doses of the current Pfizer-BioNTech COVID-19 vaccine, using a pseudovirus neutralization test (pVNT). The sera were collected from subjects 3 weeks after receiving the second dose or one month after receiving the third dose of the Pfizer-BioNTech COVID-19 vaccine. Each serum was tested simultaneously for its neutralizing antibody titer against the wild-type SARS-Cov-2 spike protein, and the Omicron spike variant. The third dose significantly increased the neutralizing antibody titers against the Omicron strain spike by 25-fold. Neutralization against the Omicron variant after three doses of the Pfizer-BioNTech COVID-19 vaccine was comparable to the neutralization against the wild-type strain observed in sera from individuals who received two doses of the companies’ COVID-19 vaccine: The geometric mean titer (GMT) of neutralizing antibody against the Omicron variant measured in the samples was 154 (after three doses), compared to 398 against the Delta variant (after three doses) and 155 against the ancestral strain (after two doses). Data on the persistence of neutralizing titers over time after a booster dose of BNT162b2 against the Omicron variant will be collected.
The Pfizer-BioNTech COVID-19 vaccine, which is based on BioNTech’s proprietary mRNA technology, was developed by both BioNTech and Pfizer. BioNTech is the Marketing Authorization Holder in the United States, the European Union, the United Kingdom, Canada and other countries and the holder of emergency use authorizations or equivalents in the United States (jointly with Pfizer) and other countries. Submissions to pursue regulatory approvals in those countries where emergency use authorizations or equivalent were initially granted are planned.

