
Category Archives: Evidence
Cartoon – Evidence Based Decision Making
Searching for value in a sea of health apps

The explosion of apps, wearables, and other health tech solutions targeted at employers has overwhelmed and frustrated many HR executives who make decisions about employee health benefits. At a recent convening of health insurance brokers we participated in, several bemoaned the challenge of helping their clients understand which solutions might bring real value.
One shared, “For the past few years, it’s felt like ‘App-apalooza’ out there. CHROs [chief human resource officers] get pitches for new apps every day…there are literally thousands out there saying they’ll reduce costs and improve employee health, but it’s next to impossible to tell which ones of them actually work.”
Brokers expressed surprise at how little evidence, or in some cases, actual patient and client experience, some health tech companies brought to the table: “We have startups coming to our clients talking about their millions of dollars in funding, but when you dig into what they’re actually doing, not only can they not show outcomes data, you find out they’ve only worked with a few dozen patients!”
But among the sea of apps purporting to manage any and every employee health need, from chronic disease to fertility to sleep quality, brokers reported their clients were finding value in a few distinct areas.
Technology-based mental health solutions received high marks for increasing access to care, with the prediction that “tele-behavioral health could become a standard part of most benefits packages very quickly”.
More surprisingly, employers shared positive feedback on the impact of virtual physical therapy solutions: “I was skeptical that it would work, but people like being able to rehab at home. And not only is it cheaper, we’re seeing higher adherence rates.”
But even the best apps are often challenged by a lack of connectivity to the rest of a patient’s healthcare. The technologies that will have the greatest staying power will be those that not only deliver results, but are able to move beyond point solutions to become part of an integrated care experience, meaningfully connected to other providers involved in a patient’s care.
Do You Really Need 10,000 Steps a Day?
Cartoon – State of the Union (Unvaccinated)
The First Sign of Civilization

Years ago, anthropologist Margaret Mead was asked by a student what she considered to be the first sign of civilization in a culture.
The student expected Mead to talk about fishhooks or clay pots or grinding stones. But no. Mead said that the first sign of civilization in an ancient culture was a femur (thighbone) that had been broken and then healed.
Mead explained that in the animal kingdom, if you break your leg, you die. You cannot run from danger, get to the river for a drink or hunt for food. You are meat for prowling beasts. No animal survives a broken leg long enough for the bone to heal.
A broken femur that has healed is evidence that someone has taken time to stay with the one who fell, has bound up the wound, has carried the person to safety and has tended the person through recovery. Helping someone else through difficulty is where civilization starts, Mead said.
“We are at our best when we serve others. Be civilized.”- Ira Byock.
Benjamin Franklin’s fight against a deadly virus: Colonial America was divided over smallpox inoculation, but he championed science to skeptics

Exactly 300 years ago, in 1721, Benjamin Franklin and his fellow American colonists faced a deadly smallpox outbreak. Their varying responses constitute an eerily prescient object lesson for today’s world, similarly devastated by a virus and divided over vaccination three centuries later.
As a microbiologist and a Franklin scholar, we see some parallels between then and now that could help governments, journalists and the rest of us cope with the coronavirus pandemic and future threats.
Smallpox strikes Boston
Smallpox was nothing new in 1721. Known to have affected people for at least 3,000 years, it ran rampant in Boston, eventually striking more than half the city’s population. The virus killed about 1 in 13 residents – but the death toll was probably more, since the lack of sophisticated epidemiology made it impossible to identify the cause of all deaths.
What was new, at least to Boston, was a simple procedure that could protect people from the disease. It was known as “variolation” or “inoculation,” and involved deliberately exposing someone to the smallpox “matter” from a victim’s scabs or pus, injecting the material into the skin using a needle. This approach typically caused a mild disease and induced a state of “immunity” against smallpox.
Even today, the exact mechanism is poorly understood and not much research on variolation has been done. Inoculation through the skin seems to activate an immune response that leads to milder symptoms and less transmission, possibly because of the route of infection and the lower dose. Since it relies on activating the immune response with live smallpox variola virus, inoculation is different from the modern vaccination that eradicated smallpox using the much less harmful but related vaccinia virus.
The inoculation treatment, which originated in Asia and Africa, came to be known in Boston thanks to a man named Onesimus. By 1721, Onesimus was enslaved, owned by the most influential man in all of Boston, the Rev. Cotton Mather.

Known primarily as a Congregational minister, Mather was also a scientist with a special interest in biology. He paid attention when Onesimus told him “he had undergone an operation, which had given him something of the smallpox and would forever preserve him from it; adding that it was often used” in West Africa, where he was from.
Inspired by this information from Onesimus, Mather teamed up with a Boston physician, Zabdiel Boylston, to conduct a scientific study of inoculation’s effectiveness worthy of 21st-century praise. They found that of the approximately 300 people Boylston had inoculated, 2% had died, compared with almost 15% of those who contracted smallpox from nature.
The findings seemed clear: Inoculation could help in the fight against smallpox. Science won out in this clergyman’s mind. But others were not convinced.
Stirring up controversy
A local newspaper editor named James Franklin had his own affliction – namely an insatiable hunger for controversy. Franklin, who was no fan of Mather, set about attacking inoculation in his newspaper, The New-England Courant.

One article from August 1721 tried to guilt readers into resisting inoculation. If someone gets inoculated and then spreads the disease to someone else, who in turn dies of it, the article asked, “at whose hands shall their Blood be required?” The same article went on to say that “Epidemeal Distempers” such as smallpox come “as Judgments from an angry and displeased God.”
In contrast to Mather and Boylston’s research, the Courant’s articles were designed not to discover, but to sow doubt and distrust. The argument that inoculation might help to spread the disease posits something that was theoretically possible – at least if simple precautions were not taken – but it seems beside the point. If inoculation worked, wouldn’t it be worth this small risk, especially since widespread inoculations would dramatically decrease the likelihood that one person would infect another?
Franklin, the Courant’s editor, had a kid brother apprenticed to him at the time – a teenager by the name of Benjamin.
Historians don’t know which side the younger Franklin took in 1721 – or whether he took a side at all – but his subsequent approach to inoculation years later has lessons for the world’s current encounter with a deadly virus and a divided response to a vaccine.
Independent thought
You might expect that James’ little brother would have been inclined to oppose inoculation as well. After all, thinking like family members and others you identify with is a common human tendency.
That he was capable of overcoming this inclination shows Benjamin Franklin’s capacity for independent thought, an asset that would serve him well throughout his life as a writer, scientist and statesman. While sticking with social expectations confers certain advantages in certain settings, being able to shake off these norms when they are dangerous is also valuable. We believe the most successful people are the ones who, like Franklin, have the intellectual flexibility to choose between adherence and independence.
Truth, not victory

What happened next shows that Franklin, unlike his brother – and plenty of pundits and politicians in the 21st century – was more interested in discovering the truth than in proving he was right.
Perhaps the inoculation controversy of 1721 had helped him to understand an unfortunate phenomenon that continues to plague the U.S. in 2021: When people take sides, progress suffers. Tribes, whether long-standing or newly formed around an issue, can devote their energies to demonizing the other side and rallying their own. Instead of attacking the problem, they attack each other.
Franklin, in fact, became convinced that inoculation was a sound approach to preventing smallpox. Years later he intended to have his son Francis inoculated after recovering from a case of diarrhea. But before inoculation took place, the 4-year-old boy contracted smallpox and died in 1736. Citing a rumor that Francis had died because of inoculation and noting that such a rumor might deter parents from exposing their children to this procedure, Franklin made a point of setting the record straight, explaining that the child had “receiv’d the Distemper in the common Way of Infection.”
Writing his autobiography in 1771, Franklin reflected on the tragedy and used it to advocate for inoculation. He explained that he “regretted bitterly and still regret” not inoculating the boy, adding, “This I mention for the sake of parents who omit that operation, on the supposition that they should never forgive themselves if a child died under it; my example showing that the regret may be the same either way, and that, therefore, the safer should be chosen.”
A scientific perspective
A final lesson from 1721 has to do with the importance of a truly scientific perspective, one that embraces science, facts and objectivity.
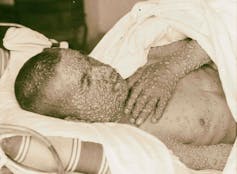
Inoculation was a relatively new procedure for Bostonians in 1721, and this lifesaving method was not without deadly risks. To address this paradox, several physicians meticulously collected data and compared the number of those who died because of natural smallpox with deaths after smallpox inoculation. Boylston essentially carried out what today’s researchers would call a clinical study on the efficacy of inoculation. Knowing he needed to demonstrate the usefulness of inoculation in a diverse population, he reported in a short book how he inoculated nearly 300 individuals and carefully noted their symptoms and conditions over days and weeks.
The recent emergency-use authorization of mRNA-based and viral-vector vaccines for COVID-19 has produced a vast array of hoaxes, false claims and conspiracy theories, especially in various social media. Like 18th-century inoculations, these vaccines represent new scientific approaches to vaccination, but ones that are based on decades of scientific research and clinical studies.
We suspect that if he were alive today, Benjamin Franklin would want his example to guide modern scientists, politicians, journalists and everyone else making personal health decisions. Like Mather and Boylston, Franklin was a scientist with a respect for evidence and ultimately for truth.
When it comes to a deadly virus and a divided response to a preventive treatment, Franklin was clear what he would do. It doesn’t take a visionary like Franklin to accept the evidence of medical science today.
Colchicine for Early COVID-19? Trial May Support Oral Therapy at Home

But some find science-by-press-release troubling.
Anti-inflammatory oral drug colchicine improved COVID-19 outcomes for patients with relatively mild cases, according to certain topline results from the COLCORONA trial announced in a brief press release.
Overall, the drug used for gout and rheumatic diseases reduced risk of death or hospitalizations by 21% versus placebo, which “approached statistical significance.”
However, there was a significant effect among the 4,159 of 4,488 patients who had their diagnosis of COVID-19 confirmed by a positive PCR test:
- 25% fewer hospitalizations
- 50% less need for mechanical ventilation
- 44% fewer deaths
If full data confirm the topline claims — the press release offered no other details, and did not mention plans for publication or conference presentation — colchicine would become the first oral drug proven to benefit non-hospitalized patients with COVID-19.
“Our research shows the efficacy of colchicine treatment in preventing the ‘cytokine storm’ phenomenon and reducing the complications associated with COVID-19,” principal investigator Jean-Claude Tardif, MD, of the Montreal Heart Institute, said in the press release. He predicted its use “could have a significant impact on public health and potentially prevent COVID-19 complications for millions of patients.”
Currently, the “tiny list of outpatient therapies that work” for COVID-19 includes convalescent plasma and monoclonal antibodies, which “are logistically challenging (require infusions, must be started very early after symptom onset),” tweeted Ilan Schwartz, MD, PhD, an infectious diseases researcher at the University of Alberta in Edmonton.
The COLCORONA findings were “very encouraging,” tweeted Martin Landray, MB ChB, PhD, of the Big Data Institute at the University of Oxford in England. His group’s RECOVERY trial has already randomized more than 6,500 hospitalized patients to colchicine versus usual care as one of the arms of the platform trial, though he did not offer any findings from that study.
“Different stage of disease so remains an important question,” he tweeted. “Maybe old drugs can learn new tricks!” Landray added, pointing to dexamethasone.
A small open-label, randomized trial from Greece had also shown less clinical status deterioration in hospitalized patients on colchicine.
“I think this is an exciting time. Many groups have been pursuing lots of different questions related to COVID and its complications,” commented Richard Kovacs, MD, immediate past-president of the American College of Cardiology. “We’re now beginning to see the fruit of those studies.”
The COLCORONA announcement came late Friday, following closely on the heels of the topline results from the ACTIVE-4a, REMAP-CAP, and ATTACC trials showing a significant morbidity and mortality advantage to therapeutic-dose anticoagulation in non-ICU patients in the hospital for COVID-19.
COLCORONA was conducted remotely, without in-person contact, with participants across Canada, the U.S., Europe, South America, and South Africa. It randomized participants double-blind to colchicine 0.5 mg or a matching placebo twice daily for the first 3 days and then once daily for the last 27 days.
Participants were ages 40 and older, not hospitalized at the time of enrollment, and had at least one risk factor for COVID-19 complications: age 70-plus, obesity, diabetes, uncontrolled hypertension, known asthma or chronic obstructive pulmonary disease, known heart failure, known coronary disease, fever of ≥38.4°C (101.12°F) within the last 48 hours, dyspnea at presentation, or certain blood cell abnormalities.
It had been planned as a 6,000-patient trial, but whether it was stopped for efficacy at a preplanned interim analysis or for some other reason was not spelled out in the press release. Whether the PCR-positive subgroup was preplanned also wasn’t clear. Key details such as confidence intervals, adverse effects, and subgroup results were omitted as well.
While a full manuscript is reportedly underway, “we don’t know enough to bring this into practice yet,” argued Kovacs.
The centuries-old drug has long been used for gout and arthritis and more recently for pericarditis along with showing promise in cardiovascular secondary prevention.
However, the drug isn’t as inexpensive in the U.S. as in Canada, Kovacs noted.
Some physicians also warned about the potential for misuse of the findings and attendant risks.
Dhruv Nayyar, MD, of the University of Toronto, tweeted that he has already had “patients inquiring why we are not starting colchicine for them. Science by press release puts us in a difficult position while providing care. I just want to see the data.”
Angela Rasmussen, MD, a virologist with the Georgetown Center for Global Health Science and Security’s Viral Emergence Research Initiative in Washington, agreed, tweeting: “When HCQ [hydroxychloroquine] was promoted without solid data, there was at least one death from an overdose. We don’t need people self-medicating with colchicine.”
As was the case with hydroxychloroquine before the papers proved little efficacy in COVID-19, Kovacs told MedPage Today: “We always get concerned when these drugs are repurposed that we might see an unintended run on the drug and lessen the supply.”
Citing the well-known diarrheal side effect of colchicine, infectious diseases specialist Edsel Salvana, MD, of the University of Pittsburgh and University of the Philippines in Manila, tweeted a plea for use only in the trial-proven patient population with confirmed COVID-19 — not prophylaxis.
The dose used was on par with that used in cardiovascular prevention and other indications, so the diarrhea incidence would probably follow the roughly 10% rate seen in the COLCOT trial, Kovacs suggested.
In the clinic, too, there are some cautions. As Elin Roddy, MD, a respiratory physician at Shrewsbury and Telford Hospital NHS Trust in England, tweeted: “Lots of drug interactions with colchicine potentially — statins, macrolides, diltiazem — we have literally been running up to the ward to cross off clarithromycin if RECOVERY randomises to colchicine.”
First Sign of Civilization

Years ago, anthropologist Margaret Mead was asked by a student what she considered to be the first sign of civilization in a culture. The student expected Mead to talk about fishhooks or clay pots or grinding stones.
But no. Mead said that the first sign of civilization in an ancient culture was a femur (thighbone) that had been broken and then healed. Mead explained that in the animal kingdom, if you break your leg, you die. You cannot run from danger, get to the river for a drink or hunt for food. You are meat for prowling beasts. No animal survives a broken leg long enough for the bone to heal.
A broken femur that has healed is evidence that someone has taken time to stay with the one who fell, has bound up the wound, has carried the person to safety and has tended the person through recovery. Helping someone else through difficulty is where civilization starts, Mead said.”
“We are at our best when we serve others. Be civilized.”
– Ira Byock.
Fauci: ‘I seriously doubt’ Russia’s coronavirus vaccine is safe and effective

Anthony Fauci, the nation’s top infectious diseases expert, said Tuesday that he has serious doubts about Russia’s announcement that it has a vaccine ready to be used for the novel coronavirus.
“Having a vaccine and proving that a vaccine is safe and effective are two different things,” Fauci said during a panel discussion with National Geographic.
The comments came just hours after Russian President Vladimir Putin said that the country had become the first in the world to gain regulatory approval for a COVID-19 vaccine.
Putin said that the vaccine went through clinical testing and that it had proven to offer immunity to the deadly disease, which has infected more than 20 million people worldwide, according to a Johns Hopkins University database.
However, phase three trials for the drug have reportedly not been completed, triggering skepticism from international health experts about its usefulness.
Fauci, the director of the National Institute of Allergy and Infectious Diseases and a key member of the White House coronavirus task force, said that he had seen no evidence supporting Putin’s position.
“I hope that the Russians have actually, definitively proven that the vaccine is safe and effective. I seriously doubt that they’ve done that,” he said, adding that Americans need to understand that the process for gaining vaccine approval requires safety and efficacy.
More than 100 possible vaccines are being developed around the world as part of efforts to offer immunity protection for the coronavirus. Moderna, in collaboration with the National Institutes of Health, launched a phase three trial for a vaccine in July, making it the first U.S. candidate to reach that stage.
Fauci has said that he’s “cautiously optimistic” that a COVID-19 vaccine will be ready by the end of the year. He told a House committee on July 31 that he was encouraged by everything he’s seen in the early data but that “there’s never a guarantee that you’re going to get a safe and effective vaccine.”
The World Health Organization said Tuesday that it was monitoring Russia’s progress in developing a COVID-19 vaccine. Progress in combating the virus “should not compromise safety,” the health agency said.
Former Food and Drug Administration Commissioner Scott Gottlieb echoed Fauci’s skepticism earlier Tuesday, noting in a tweet that Russia has been behind disinformation campaigns related to the pandemic.
“Today’s news that they ‘approved’ a vaccine on the equivalent of phase 1 data may be another effort to stoke doubts or goad U.S. into forcing early action on our vaccines,” he said.
Russia is reportedly planning to offer its COVID-19 vaccine to medical personnel as soon as this month. It will be made available to the general public in October, according to Reuters.



