
Category Archives: Physician
DOJ Questions UnitedHealth Doctors Re: Medicare Advantage Upcoding

I’ve been at this for so long and have seen so much. And it’s hard to overstate how significant the latest revelations from The Wall Street Journal are. According to its reporting, the U.S. Department of Justice’s criminal health care-fraud unit is questioning former UnitedHealth Group employees about the company’s Medicare billing practices regarding how the company records diagnoses that trigger higher payments from taxpayers.
For years, independent policy experts and *some* regulators have warned that the private Medicare Advantage program has become a breeding ground for upcoding and tax dollar waste. The tactic being scrutinized by the DOJ is called “upcoding.” Essentially, Medicare Advantage companies have an incentive to “find” new illnesses — even among patients who might not need additional treatment because the more serious the diagnoses, the bigger the government payouts to the company.
According to the Journal, prosecutors, FBI agents, and the Health and Human Services Inspector General have been asking ex-employees about special training for doctors, software that flags profitable conditions, and even bonuses for physicians who recode patient files. One former UnitedHealth doctor told the Journal that prosecutors inquired about pressure to use certain diagnosis codes and bonus pay for certain health care decisions that financially favored UnitedHealth.
The Journal’s data shows that UnitedHealth’s members received certain lucrative diagnoses at higher rates than patients in other Medicare Advantage plans — billions of extra dollars that ultimately come from taxpayers. In one example, they reportedly pulled in about $2,700 more taxpayer dollars per patient visit when nurses went into seniors’ homes to hunt for additional conditions.
In a statement, UnitedHealth insists they “remain focused on what matters most: delivering better outcomes, more benefits, and lower costs for the people we serve.”
This latest criminal investigation joins at least two other DOJ probes into UnitedHealth’s billing and potential antitrust violations. And it’s yet another reminder that the Medicare Advantage program — which, much to many advocates alarm, now covers more than half of all Medicare enrollees – is desperately in need of real oversight.
If there’s any silver lining, it’s that courageous former employees are speaking up. They know what I know: This “profit-maximizing” through “upcoding” and “favorable selection” drains billions that could be better spent on actual patient care and pad Wall Street profits.
Poll results: AGI and the future of medicine

Artificial general intelligence (AGI) refers to AI systems that can match or exceed human cognitive abilities across a wide range of tasks, including complex medical decision-making.
With tech leaders predicting AGI-level capabilities within just a few years, clinicians and patients alike may soon face a historic inflection point: How should these tools be used in healthcare, and what benefits or risks might they bring? Last month’s survey asked your thoughts on these pressing questions. Here are the results:

My thoughts:
I continue to be impressed by the expertise of readers. Your views on artificial general intelligence (AGI) closely align with those of leading technology experts. A clear majority believes that AGI will reach clinical parity within five years. A sizable minority expect it will take longer, and only a small number doubt it will ever happen.
Your answers also highlight where GenAI could have the greatest impact. Most respondents pointed to diagnosis (helping clinicians solve complex or uncertain medical problems) as the No. 1 opportunity. But many also recognized the potential to empower patients: from improving chronic disease management to personalizing care. And unlike the electronic health record, which adds to clinicians’ workloads (and contributes to burnout), GenAI is widely seen by readers as a tool that could relieve some of that burden.
Ultimately, the biggest concern may lie not with the technology, itself, but in who controls it. Like many of you, I worry that if clinicians don’t lead the way, private equity and for-profit companies will. And if they do, they will put revenue above the interests of patients and providers.
Thanks to those who voted. To participate in future surveys, and for access to timely news and opinion on American healthcare, sign up for my free (and ad-free) newsletter Monthly Musings on American Healthcare.
* * *
Dr. Robert Pearl is the former CEO of The Permanente Medical Group, the nation’s largest physician group. He’s a Forbes contributor, bestselling author, Stanford University professor, and host of two healthcare podcasts. Check out Pearl’s newest book, ChatGPT, MD: How AI-Empowered Patients & Doctors Can Take Back Control of American Medicine with all profits going to Doctors Without Borders.
Cartoon – Medicare Advantage Office Visit
CMS proposes a nearly 3% cut to Medicare physician pay for 2025
https://www.kaufmanhall.com/insights/blog/gist-weekly-july-12-2024

On Wednesday, the Centers for Medicare & Medicaid Services (CMS) issued its proposed annual changes to physician payments in its 2025 Medicare Physician Fee Schedule Proposed Rule. Required by statute to maintain budget neutrality, CMS is proposing to reduce the conversion factor—which translates the cost of providing medical services into physician payments—by 2.8%.
This is expected to lower physician payments by 2.93% on average. The proposed rule also includes new telehealth flexibilities, changes allowing eligible accountable care organizations access to a quarterly advance on their earned savings, and new payments for providers that help patients at high risk of overdose or suicide.
The Gist: With CMS proposing to reduce physician payments for the fifth straight year, the American Medical Association and other physician groups are once again calling on Congress to avert these cuts.
Congress has previously responded with “Band-Aid” solutions to temporarily reduce or eliminate reductions for the next calendar year, but physician groups are demanding a more comprehensive fix that ties Medicare payment updates to the Medicare Economic Index, a measure of practice cost inflation.
Medicare physician pay has declined 20% relative to practice costs from 2000 to 2021, and post-COVID inflation has only worsened the issue. Although lawmakers on Capitol Hill have explored various means of doing so, structural changes to Medicare budgetary policy face an uphill legislative battle in a presidential election year.
Medicare Physician Payment Not Keeping Up
https://www.kaufmanhall.com/insights/infographic/medicare-physician-payment-not-keeping
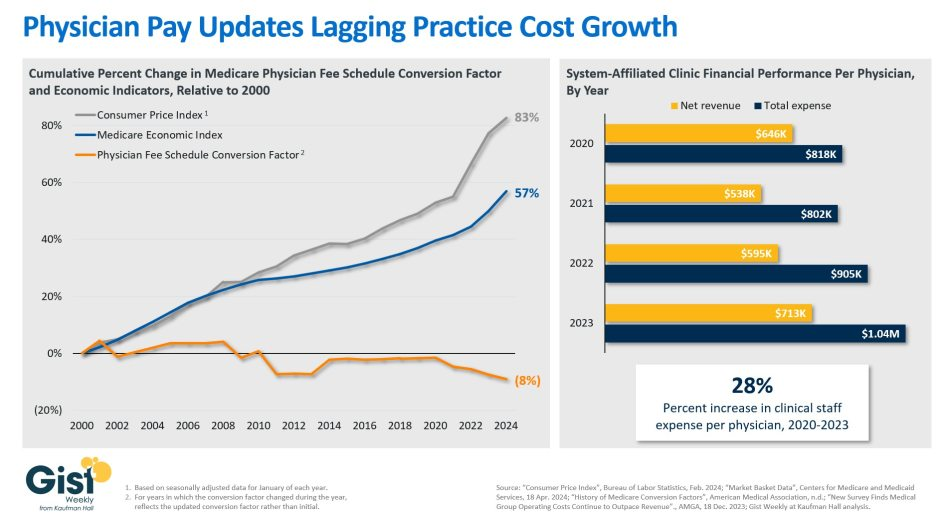
Following the US Senate Finance Committee’s recent white paper on Medicare physician payment reform, the graphic above shows how Medicare payments to physicians have not kept pace with inflation.
The Medicare physician fee schedule conversion factor, which is used to assign dollar amounts to relative value units, decreased by eight percent between 2000 and 2024.
Over the same period, the Medicare Economic Index (MEI), which measures practice cost inflation, and Consumer Price Index rose 57 percent and 83 percent, respectively. Reductions to the conversion factor contributed to a 20 percent decline in Medicare physician pay relative to practice cost inflation from 2000 to 2021.
Physician practice margins in general have also worsened since COVID, not just due to Medicare payments.
Although net revenue per physician at system-affiliated clinics increased about 10 percent from 2020 to 2023,total expenses per physician rose about 27 percent, driven by a per physician clinical staffing expense increase of nearly the same amount.
Both the Senate Finance Committee’s white paper and a recently introduced House bill suggest tying the Medicare physician fee schedule conversion factor updates to the MEI, either partially or fully, but immediate legislative action in an election year appears unlikely.
In the meantime, physician practices continue to face a difficult operating environment with costs rising faster than revenues.
Handicapping the Players in the Quest for Healthcare Affordability

As campaigns for November elections gear up for early voting and Congress considers bipartisan reforms to limit consolidation and enhance competition in U.S. healthcare, prospective voters are sending a cleat message to would-be office holders:
Healthcare Affordability must be addressed directly, transparently and now.
Polling by Gallup, Kaiser Family Foundation and Pew have consistently shown healthcare affordability among top concerns to voters alongside inflation, immigration and access to abortion. It is higher among Democratic-leaning voters but represents the majority in every socio-economic cohort–young and old, low and middle income and households with/without health insurance coverage., urban and rural and so on.
It’s understandable: household economic security is declining: per the Federal Reserve’s latest household finances report:
- 72% of US adults say they are doing well financially (down from 78% in 2021)
- 54% say they have emergency savings to cover 3 months expenses ($400)—down from high of 59% in 2015.
- 69% say their finances deteriorated in 2023. They’re paying more for groceries, fuel, insurance premiums and childcare.
- Renters absorbed a 10% increase last year and mortgage interest spike has put home ownership beyond reach for 6 in 10 households
Thus, household financial security is the issue and healthcare expenses play a key role. Drug prices, hospital consolidation, price transparency and corporate greed will get frequent recognition in candidate rhetoric. “Reform” will be promised. And each sector in the industry will offer solutions that place the blame on others.
Granted, the U.S. health system lacks a uniform definition of healthcare affordability. It’s a flaw. In the Affordable Care Act, it was framed in the context of an individual’s eligibility for government-subsidized insurance coverage (8.39% adjusted gross income for households between 100% and 400% of the federal poverty level). But a broader application to the entire population was overlooked. Nonetheless, economists, regulators and consumers recognize the central role healthcare affordability plays in household financial security.
Handicapping the major players potential to win the hearts and minds of voters about healthcare affordability is tricky:
- Each major sector has seen the ranks of its membership decrease and the influence (and visibility) of its bigger players increase. They’re easy targets for industry critics.
- Each sector is seeing private equity and non-traditional players play bigger roles. The healthcare landscape is expanding beyond the traditional players.
- Each sector is struggling to make their cases for incremental reforms while employers, legislators and consumers want more. Bipartisan support for anything is a rarity: an exception is antipathy toward healthcare consolidation and lack of price transparency.
- All recognize that affordability is complicated. Unit cost and price increases for goods and services are the culprit: excess utilization is secondary.
Against this backdrop, here’s a scorecard on the current state of preparedness as each navigates affordability going into Campaign 2024:
| Sector | Advantages | Disadvantages | Handicap Score1=Unprepared to5=Well Prepared |
| Hospitals | Community presence (employer, safety net) Economic impact Influence in Congress Scale: 30% of spending + direct employment of 52% of physicians Access to capital | Lack of costs & price transparency Unit costs inflation due to wage, supply chain & admin Shifting demand for core services. Low entry barriers for key services Regulator headwind (state, federal). Operating, governing culture Value proposition erosion with employers, pre-Medicare populations Consumer orientation | 3 |
| Physicians | Consumer trust Influence in Congress Shared savings (Medicare) Essentiality Specialization Access to technology | Care continuity Inadequacy of primary care Disorganization (fragmentation) Value of shared savings to general population (beyond Medicare) Culture: change-averse (education, licensing performance measurement, et al) Data: costs, outcomes | 2 |
| Drug Manufacturers | Increasing product demand Influence in Congress Public trust in drug efficacy Insurance structure that limits consumer price sensitivity to OOP Potential for AI -enabled discovery, market access Access to private capital Congress’ constraint on PBMs | Unit cost escalation Lack of price transparency Growing disaffection for FDA Long-term Basic Research Funding State Price Control Momentum Market access Restrictive Formulary Growth Transparency in Distributor-PBM business relationships Public perception of corporate greed | 2 |
| Health Insurers | Availability of claims, cost data Employer tax exemptions Growing government market Plan design: OOP, provider access Public association: coverage = financial security Access to private capital | Escalating premiums Declining group market Growing regulatory scrutiny (consolidation, data protection) Tension with health systems Value proposition erosion among government, employers, consumers | 4 |
| Retail Health | Non-incumbrance of restrictive regulatory framework Consumer acceptance Breadth of product opportunities Access to private capital Opportunity for care management (i.e. CVS- Epic) Operational orientation to consumers (convenience, pricing, et al) Potential with employers, | Lack of access, coordination with needed specialty care Threat of regulatory restraint on growth Risks associated with care management models | 3 |
The biggest, investor-owned health insurers own the advantage today. As in other sectors, they’re growing faster than their smaller peers and enjoy advantages of scale and private capital access to fund their growth. A handful of big players in the other sectors stand-out, but their affordability solutions are, to date, not readily active.
In each sector above, there is consensus that a fundamental change in the structure, function and oversight of the U.S. health is eminent. In all, tribalism is an issue: publicly-owned, not for profits vs. investor-owned, independent vs. affiliated, big vs. small and so on.
Getting consensus to address affordability head on is hard, so not much is done by the sectors themselves. And none is approaching the solution in its necessary context—the financial security of a households facing unprecedented pressures to make ends meet. In all likelihood, the bigger, more prominent organizations in their ranks of these sectors will deliver affordability solutions well-above the lowest common denominators that are comfortable for most Thus, health care affordability will be associated with organizational brands and differentiated services, not the sectors from which their trace their origins. And it will be based on specified utilization, costs, outcome and spending guarantees to consumers and employers that are reasonable and transparent.
Healthcare winners and losers after FTC bans non-compete clauses

With a single ruling, the Federal Trade Commission removed the nation’s occupational handcuffs, freeing almost all U.S. workers from non-compete clauses. The medical profession will never be the same.
On April 23, the FTC issued a final rule, affecting not only new hires but also the 30 million Americans currently tethered to non-compete agreements. Scheduled to take effect in September—subject to the outcome of legal challenges by the U.S. Chamber of Commerce and other business groups—the ruling will dismantle longstanding barriers that have kept healthcare professionals from changing jobs.
The FTC projects that eliminating these clauses will boost medical wages, foster greater competition, stimulate job creation and reduce health expenditures by $74 billion to $194 billion over the next decade. This comes at a crucial moment for American healthcare, an industry in which 60% of physicians report burnout and 100 million people (41% of U.S. adults) are saddled with medical bills they cannot afford.
Like all major rulings, this one creates clear winners and losers—outcomes that will reshape careers and, potentially, alter the very structure of U.S. healthcare.
Winners: Newly Trained Clinicians
Undoubtedly, the FTC’s ruling is a win for younger doctors and nurses, many of whom join hospitals and health systems with the promise of future salary increases and more autonomy. However, by agreeing to stringent non-compete clauses, these newly trained clinicians have little choice but to place their trust in employers that, shielded by air-tight agreements, have no fear of breaking their promises.
Most newly trained clinicians enter the medical job market in their late 20s and early 30s, carrying significant student-loan debt—nearly $200,000 for the average doctor. Eager for stable, well-paying positions, these young professionals quickly settle into their careers and communities, forming strong relationships with friends and patients. Many start families.
But when these clinicians realize their jobs are falling short of the promises made early on, they face a tough decision: either endure subpar working conditions or uproot their lives. Taking a new job 25 or 50 miles away or moving to a different state are often are only options to avoid breaching a non-compete clause.
In a 570-page supplement to its ruling, the FTC published testimonials from dozens of healthcare professionals whose lives and careers were harmed by these clauses.
“Healthcare providers feel trapped in their current employment situation, leading to significant burnout that can shorten their career longevity,” said one physician working in rural Appalachia.
By banning non-competes, the FTC’s rule will boost career mobility for all clinicians within their own communities. This change will likely spur competition among employers—leading to improved pay and benefits to attract and, equally important, retain top talent. And with the reassurance that they can easily switch jobs if their current employer falls short of expectations, clinicians will enjoy greater professional satisfaction and less burnout.
Winners: Patients In Competitive Markets
Benefits that accrue to doctors and nurses from the FTC’s ban will translate directly to improved outcomes for patients. For example, we know that physicians who report symptoms of burnout are twice as likely to commit a serious medical error. Studies have shown the inverse is true, as well: healthcare providers who are satisfied with their jobs tend to have lower burnout rates, which is positively correlated with improved patient outcomes.
Once freed from restrictive non-compete clauses, many clinicians will practice elsewhere within the community. To attract patients, they will have to offer greater access, lower prices and more personalized service. Others with the freedom to choose will join outpatient centers that offer convenient and efficient alternatives for diagnostic tests, surgery and urgent medical care, often at a fraction of the cost of traditional hospital services. In both cases, increased competition will give patients improved medical care and added value.
Losers: Large Health Systems
Large health systems, which encompass several hospitals in a geographic area, have traditionally relied on non-compete agreements to maintain their market dominance. By barring high-demand medical professionals such as radiologists and anesthesiologists from joining competitors or starting independent practices, these systems have been able to suppress competition and force insurers to pay more for services.
Currently, these systems can demand high reimbursement rates from insurers while also maintaining relatively low wages for staff, creating a highly profitable model. Yale economist Zack Cooper’s research shows the consequence of the status quo: prices go up and quality declines in highly concentrated hospital markets.
The FTC’s ruling challenges those conditions, potentially dismantling monopolistic market controls. As a result, insurers will no longer be forced to contend with a single, dominant provider. And with health systems pushed to offer better wages and benefits to retain their top talent, bottom lines will shrink.
While nonprofit hospitals and health systems are not currently under the FTC’s jurisdiction, the agency has pointed out that these facilities might be at “a self-inflicted disadvantage in their ability to recruit workers.” Moreover, as Congress intensifies scrutiny on the nonprofit status of U.S. health systems, hospitals that do not voluntarily align with the FTC’s guidelines may find themselves compelled to do so through legislative actions.
Losers: Hospital Administrators
Individual hospitals have faced a unique challenge over the past decade. Across the country, inpatient numbers are falling, which makes it harder for hospital administrators to fill beds overnight. This trend has been driven by advancements in medical technology and new practices that enable more outpatient procedures, along with changes in insurance reimbursements favoring less costly outpatient care. As a result, hospital administrators have been compelled to adapt their financial strategies.
Nowadays, outpatient services account for about half of all hospital revenue. These range from physician consultations to specialized procedures like radiological and cardiac diagnostics, chemotherapy and surgeries.
Medicare and other insurers typically pay hospitals more for these outpatient services than they pay local doctors and other facilities. Knowing this, hospitals are hiring community doctors and acquiring diagnostic and procedural facilities, then boosting profitability by charging the higher hospital rates for the same services.
Hospital administrators know that this strategy only works if the newly hired clinicians are prohibited from quitting and returning to practice within the same community. If they do, their patients are likely to go with them. This is why the non-compete clauses are so essential to a hospital’s financial success.
As expected, the American Hospital Association opposes the FTC’s rule, arguing that non-compete clauses protect proprietary information. In practice, most of the doctors affected by the ban are providing standard medical care and have no proprietary knowledge that requires protection.
Looking Ahead
Today’s hospital systems are starkly divided between haves and have-nots. Facilities in affluent areas often enjoy high reimbursement rates from private insurers, boosting financial success and administrator salaries. In contrast, rural hospitals grapple with low patient volumes while facilities in economically disadvantaged, high-population areas face greater financial difficulties.
The current model is not working. The old ways of doing things—enforcing non-competes, charging higher fees for identical services and promoting market consolidation to hike prices—are not sustainable solutions.
The abolition of non-compete agreements will produce both winners and losers. In the healthcare sector, the ultimate measure of a policy’s impact should be its effect on patients—and the overwhelming evidence suggests that eliminating these clauses will benefit them greatly.
The Do’s and Don’ts of Navigating the Health System when you Need It: My First-Hand Experience

I fell down a flight of stairs at 4 a.m. last Wednesday.
It was totally my fault.
Since then, I have used hospital emergency departments in 2 states, a freestanding imaging center and a large orthopedic clinic and I’m just getting started. Six days in, I’m lucky to be alive but I still don’t know the extent of my injuries, my chances of playing golf again nor what I will end up spending on this ordeal. But nonetheless, it could have been worse. I’m alive.
Surprises in all aspects of life are never anticipated fully and always disruptive. This one, for me, is no exception. I am frustrated by my accident and uncomfortable with sudden dependence on others to help navigate my recovery.
But this is also a teachable moment., As I am navigating through this ordeal, I find myself reflecting on the system—how it works or doesn’t—based on what I am experiencing as a patient.
Here’s my top three observations thus far:
The patient experience is defined by the support team:
The heroes in every setting I’ve used are the clerks, technicians, nurses and support staff who’ve made the experiences tolerable and/or reassuring. Patients like me are scared. Emotional support is key: some of that is defined by standard operating procedures and checklists but, in other settings, it’s cultural. Genuineness, empathy and personal attention is easy to gauge when pain is a factor. By the time physicians are on the scene, reassurance or fear is already in play. Care teams include not just those who provide hands-on care, but the administrative clerks and processes that either heighten patient anxiety or lessen fear. The health and well-being of the entire workforce—not just those who deliver hands-on care—matters. And it’s easy to see distinctions between organizations that embrace that notion and those that don’t.
Navigation is no-man’s land:
The provider organizations I’ve used thus far have 3 different owners and 3 different EHR systems. Each offers written counsel about ‘patient responsibility’ and each provides a list of do’s and don’ts for each phase of the process. Sharing test results across the 3 provider organizations is near impossible and coordination of care management is problematic unless all parties agree and protocols facilitating sharing in place. Perhaps because it was a holiday weekend, perhaps because staffing levels were less than usual, or perhaps because the organizations are fierce competitors, navigating the system has been unusually difficult. Navigating the system in an emergency is essential to optimal outcomes: processes to facilitate patient navigation are not in place.
What’s clear is hospitals, clinics and imaging facilities on different EHR systems don’t exchange data willingly or proactively. And, at every step, getting approvals from insurers a major step in the processes of care.
Price transparency is a non-issue in emergency care:
The services I am receiving include some that are “shoppable” and many that aren’t. I have no idea what I will end up spending, my out-of-pocket obligations nor what’s to come. I know among the mandatory forms I signed in advance of treatment in all 3 sites were consent forms for treatment and my obligation for payment. But in an emergency, it’s moot: there’s no way to know what my costs will be or my out-of-pocket responsibility. So, the hospital and insurer price transparency rules (2021, 2022) might elevate awareness of price distinctions across settings of care but their potential to bend the cost curve is still suspect.
Patients, like me, have to fend for ourselves. I am a number. Last Wednesday, waiting 85 minutes to be seen was frightening and frustrating though comparatively fast. Duplicative testing, insurer approvals, work-shift transitions, bedside manners, team morale, and sterile care settings seem the norm more than exception.
So, for me, the practical takeaways thus far are these:
- Don’t have an accident on a holiday weekend.
- Don’t expect front desk and check-out personnel to engage or answer questions. They’re busy.
- Don’t expect to start or leave without paying something or agreeing you will.
- Don’t expect waiting areas and exam rooms to be warm or inviting.
- Do have great neighbors and family members who can help. For me, Joe, Jordan, Erin and Rhonda have been there.
The health system is complicated and relationships between its major players are tense. Not surprisingly and for many legitimate reasons, my experience, thus far, is the norm. We can do better.
Paul
P.S. As I have reflected on the event last week, I found myself recalling the numerous times I called on “my doctors” to help my navigation of the system. They include Charles Hawes (deceased), Ben Womack, Ben Heavrin, David Maron, David Schoenfeld and Blake Garside. And, in the same context, the huge respect I have for clinicians I’ve known through Vanderbilt and Ohio State like Steve Gabbe and Andy Spickard who personify the best the medical profession has to offer. Thanks gentlemen. What you do matters beyond diagnoses and treatments. Who you are speaks volumes about the heart and soul of this industry now struggling to re-discover its purpose.
The state of state physician noncompete bans
https://www.kaufmanhall.com/insights/blog/gist-weekly-may-17-2024

With the Federal Trade Commission (FTC) issuing a final rule last month that bans noncompete agreements nationwide, the graphic above is our attempt to categorize the current status of complex state noncompete laws that affect physicians.
Except in the event of a business sale, five states—California, North Dakota, Minnesota, Nebraska, and Oklahoma—ban all noncompete agreements for all employees, and at least 19 states either ban them for physicians or place varying limits on them for physicians.
Examples of these limits include a narrow law in Florida that allows noncompetes to be voided if there is only one employer of a physician specialty in a county, and a Tennessee law that only permits physician noncompetes that bar a physician from practicing at facilities where their former employer provides services.
As a noncompete agreement can restrict a physician’s ability to practice near a former employer for years, bans on physician noncompete agreements have been shown to improve community access to care. One study found that, compared to places that allow them, places that banned noncompetes for physicians saw increased physician employment, the opening of more physician practices, and a lower likelihood of practice closures.
Should the new FTC ban survive the mounting legal challenges it faces, its effect on the physician labor market may be limited, as not-for-profit organizations fall outside the FTC’s traditional enforcement jurisdiction. However, the agency has indicated a willingness to reevaluate an entity’s not-for-profit status and stated that “some portion” of tax-exempt hospitals could fall under the final rule’s purview.


