
Cartoon – Sending Our Medical Warriors to Battle


https://mailchi.mp/39947afa50d2/the-weekly-gist-april-17-2020?e=d1e747d2d8

Blue Shield of California announced last Friday that its healthcare services division, Altais, is acquiring Brown & Toland Physicians, a multispecialty network of 2,700 physicians serving 350,000 patients in the greater San Francisco Bay Area. Brown & Toland, formed in 1993, is a clinically-integrated network of independent physicians that has received much attention nationally for its risk-based contracting as both a Medicare Pioneer Accountable Care Organization, as well for its landmark contract to manage state workers and retirees in the California Public Employees’ Retirement System (CalPERS).
While few details of the deal have been released, Altais says it will provide Brown & Toland with both capital for growth, and a technology platform that includes practice management, analytics tools, telehealth and electronic health record assistance. Brown & Toland’s CEO, Kelly Robinson, said the partnership would enable the group to expand geographically.
While Blue Shield’s purchase of Brown & Toland is the first noteworthy payer acquisition of physician practices we’ve seen in the post-COVID era, it’s likely just the first of many to follow in coming months. As we reported last week, the majority of physician groups—especially smaller independent practices—are suffering significant financial strain, which will likely make groups of all sizes more open to partnership options.
Recent reports suggest that payers in particular may be weathering the economic shocks of the crisis relatively well. This week UnitedHealth Group (UHG) announced it exceeded Q1 earnings targets, and would maintain its pre-COVID earnings guidance for the year, citing savings from cancelled routine care and elective procedures. Should payers continue to fare well, it’s likely that UHG and other health plans could enjoy an advantage in deploying the capital necessary to roll up distressed physician practices.
https://mailchi.mp/39947afa50d2/the-weekly-gist-april-17-2020?e=d1e747d2d8

Early reports of hastened recoveries among patients taking the antiviral drug remdesivir sent manufacturer Gilead Sciences’ stock soaring over 8 percent this morning, and contributing to an overall uptick in the market. The gains came after a scoop by healthcare news site STAT, which obtained a copy of an internal webinar from University of Chicago Medicine, where an infectious disease specialist discussed positive results from their early experience with remdesivir. The system recruited 125 patients into Gilead’s Phase 3 clinical trials for the drug; 113 patients had severe disease. The presenting physician reported rapid reductions in fever and improvements in respiratory symptoms, noting that just two patients had died, and most of the participating patients had already been discharged—on average after just six days, suggesting a long course of drug treatment may not be necessary.
The STAT leak comes on the heels of a NEJM article late last week, which reported clinical improvement of over two-thirds in COVID-19 patients who received remdesivir. Critics were quick to point out numerous flaws in the study, including lack of a control group, cherry-picking of patients, and the deep involvement of the manufacturer in study design, many of which also apply to the University of Chicago report.
In the thick of the pandemic, doctors and patients’ families are understandably motivated to get very sick patients access to any treatment that may help—but the resulting frenzy following the publication of early results may make it even harder to get good data to understand what works, and what doesn’t.
In the words of one expert, “Fast trials are generally not very interpretable, interpretable trials are generally not fast”. In the search for a “COVID-19 cure”, it’s highly unlikely that any single drug will provide a cure for the viral illness, and the only way we’ll know if a treatment is truly working is to wait for the results of randomized, controlled trials—despite how frustrating it is to muster the patience to do so.
https://mailchi.mp/39947afa50d2/the-weekly-gist-april-17-2020?e=d1e747d2d8

It was another brutal week in the coronavirus pandemic, with more than 2.1M cases and nearly 150,000 deaths worldwide. The US continued to be the hardest-hit country, reaching a daily record 4,591 deaths from COVID-19 on Thursday. The national death toll is now more than 35,000, though there are signs that the number of new cases in the US has begun to plateau, raising hopes that the worst days may be drawing to a close. Meanwhile, with strict stay-at-home measures continuing in most places across the country, the economic toll of the virus mounted. New unemployment claims rose by another 5.2M, bringing the estimated number of American jobs claimed by the virus to 22M, eliminating a decade’s worth of job growth, and raising the unemployment rate to an estimated 17 percent.
As the growth in new cases flattened, attention turned this week to plans to “reopen” the American economy. Despite insisting early in the week that he alone would decide when and how to reopen the country, President Trump yesterday unveiled a set of non-binding, “Opening Up America Again” guidelines for state and local officials to use in judging when to loosen restrictions. The guidelines suggest a three-stage, gated approach, gradually allowing individuals and employers to return to normal activities based on criteria including disease trends, hospital capacity, and the availability of robust testing. Progressing from one stage to the next is predicated on maintaining a downward trajectory in new cases—with any signs of a resurgence indicating a need to reimpose restrictions.
Missing from the White House plan are specific details about how states, cities, and healthcare providers are to procure and pay for the many millions of tests and extensive contact tracing that will need to be available to allow businesses, public transport systems, and other essential services to resume activity. By week’s end, about 3.5M coronavirus tests had been conducted nationally, but the daily number of tests conducted has plateaued, and the test-positivity rate is still troublingly high. Public health experts continue to warn that testing must ramp up significantly before any steps toward reopening can be considered, a difficult challenge given widespread reports of shortages of testing supplies and trained lab technicians. To bolster testing capacity, the Centers for Medicare and Medicaid Services (CMS) this week nearly doubled the amount it will pay laboratories to analyze tests using high-throughput equipment.
Three coalitions of states—in the Northeast, Midwest, and West Coast—were formed this week to coordinate regional efforts to reopen the economy. Among the issues they’ll need to address: interstate travel restrictions, coordinated purchasing of critical supplies, investments in contact tracing capabilities, and ongoing surveillance of the virus’ spread. With federal agencies taking a back seat to states (“You are going to call your own shots,” the President told governors on a call this week), it became clear that the road back from the coronavirus pandemic will be circuitous, with a patchwork of different timelines and approaches in different locations based on local conditions and resources.
In the words of William Gibson, “The future is here—it’s just not very evenly distributed.”
https://www.ft.com/coronavirus-latest
The human cost of the coronavirus outbreak has continued to mount, with more than 2.2m cases confirmed globally and more than 141,900 people known to have died from the disease. The World Health Organization has declared the outbreak a pandemic and it has spread to more than 190 countries around the world. This page provides an up-to-date visual narrative of the spread of Covid-19 so please check back regularly because we will be refreshing it with new graphics and features as the story evolves.

But the experience of 1918 also reminds us that early, layered (i.e., more than one at the same time) and lengthy mitigation measures are the best strategy. For social distancing to work, it must be sweeping and enforced across a wide swath of the community. Essential businesses will, of course, need to continue. All other places where people congregate should cease operations for the time being. In 1918, social distancing measures were kept in place for many weeks, if not months, even if people and businesses did not always support them. But the key lesson: This approach worked.
By now, many have read of the comparisons between St. Louis, where a decisive health commissioner reacted with amazing rapidity to implement sweeping public health orders, and Philadelphia, which chose to stay open, even going ahead with plans for a huge parade.
St. Louis was rewarded with one of the best outcomes of any large U.S. city. Philadelphia’s fateful decision to carry on with its immense Liberty Loan Parade resulted in a massive spike in influenza cases in the days immediately following. The city endured some of the worst numbers of cases and deaths in the United States as a result.
Philadelphia was hardly alone, however. In Baltimore, the health commissioner dragged his feet when a group of physicians requested that the city ban public gatherings. “We do not consider such drastic steps necessary in view of the extreme low civilian death rate in the city,” he told them. More than 4,100 Baltimoreans lost their lives to the epidemic.
In Atlanta, the mayor sided with business interests and reopened the city after just three weeks of closures, over the vocal objections of his Board of Health. When the board predicted that Atlanta’s epidemic peak would not occur for another nine days, the mayor dismissed the science, arguing that there was no way to foretell future conditions. The city health officer sided with the mayor, mistakenly declaring that the peak had passed. It had not, and Atlanta’s fall wave of the epidemic raged on, unchecked, through the end of 1918. “The influenza situation in Atlanta is up to the people themselves,” the Public Safety Committee declared.
Atlanta may be a more extreme example, but its experience was hardly singular. In every city we studied from this era there was public pressure to quit the social distancing measures as soon as the epidemic seemed to peak and then ebb. Thinking that the proverbial coast was clear, many communities lifted social distancing measures before the battle was truly over. After weeks of being denied their usual social outlets, people were eager to return to a life of normalcy, and they did so in one giant rush. In city after city, masses lined up for movie houses and performance theaters, crowds packed into dance halls and cabarets, and throngs flocked to downtown shopping districts, often on the very day that the closure orders were lifted.
The result? Cases and deaths resurged. Most cities closed their schools once again. But the political, economic and social will to issue another round of sweeping business closures and gathering bans had evaporated as people grew weary of the dislocations of social distancing. In some cities, most notably Denver, Kansas City, Milwaukee and even the vaunted St. Louis, this second peak was even deadlier than the first.
Lastly, 1918 teaches us how quickly an unchecked epidemic can overwhelm our health-care infrastructure. Philadelphia had to erect 32 temporary hospitals just to handle its massive number of influenza cases. On a single day in mid-October, 10 trucks were needed to carry the bodies of indigent victims to the city’s potter’s field. Some of the deceased had to be buried in temporary graves until more permanent plots could be dug.
In Pittsburgh, the epidemic grew so bad that a local sporting club had to donate its tents to use as field hospitals. One San Antonio hospital had to rely on 18 student nurses to tend to hundreds of influenza patients; the 12 regular nurses were all sick with influenza themselves. Nashville’s City Hospital was overrun with cases in a single day. These cities, unfortunately, were not alone in their experiences.
Today we have two notable advantages over those in 1918: We know the causative agent of covid-19, and our medical care is far more advanced. In 1918, scientists believed the epidemic was caused by a bacterium, and the influenza virus would not be discovered for another quarter-century. The standard medical treatment for influenza victims in 1918 consisted of little more than propping patients up to prevent them from choking on their sputum. Today, it is only a matter of time before researchers discover pharmaceutical therapies and develop an effective vaccine against the disease. In 2020, physicians have the ability to drive down the fatality rate of this epidemic through the use ventilators and intensive care units — as long as such lifesaving machines are available.
Our health-care system can only do this, however, if we don’t allow our already-taxed hospitals, physicians and nurses to be overrun with cases. That means that, until an effective vaccine can be developed and deployed, we must “flatten the curve.” This will not be accomplished in a week, or even a month. We must implement and coordinate sweeping non-pharmaceutical interventions on a national level and keep these measures in place as long as necessary. These measures are not perfect. They are slow and plodding. They are socially and economically disruptive. They fracture the routines of our daily lives in myriad ways, large and small. They do not magically end epidemics. But they can save lives.
As we all endure the hardships of the covid-19 pandemic and dislocations of social distancing, we can take heart that together we will save lives. Just as our forebears did a century ago.
And that is the most important lesson of 1918.

Few figures tell you anything useful about how the coronavirus has spread through the U.S. Here’s one that does.
How many people have the coronavirus in the United States? More than two months into the country’s outbreak, this remains the most important question for its people, schools, hospitals, and businesses. It is also still among the hardest to answer. At least 630,000 people nationwide now have test-confirmed cases of COVID-19, according to The Atlantic’s COVID Tracking Project, a state-by-state tally conducted by more than 100 volunteers and experts. But an overwhelming body of evidence shows that this is an undercount.
Whenever U.S. cities have tested a subset of the general population, such as homeless people or pregnant women, they have found at least some infected people who aren’t showing symptoms. And, as ProPublica first reported, there has been a spike in the number of Americans dying at home across the country. Those people may die of COVID-19 without ever entering the medical system, meaning that they never get tested.
There is clearly some group of Americans who have the coronavirus but who don’t show up in official figures. Now, using a statistic that has just become reliable, we can estimate the size of that group—and peek at the rest of the iceberg.
According to the Tracking Project’s figures, nearly one in five people who get tested for the coronavirus in the United States is found to have it. In other words, the country has what is called a “test-positivity rate” of nearly 20 percent.
That is “very high,” Jason Andrews, an infectious-disease professor at Stanford, told us. Such a high test-positivity rate almost certainly means that the U.S. is not testing everyone who has been infected with the pathogen, because it implies that doctors are testing only people with a very high probability of having the infection. People with milder symptoms, to say nothing of those with none at all, are going undercounted. Countries that test broadly should encounter far more people who are not infected than people who are, so their test-positivity rate should be lower.
The positivity rate is not the same as the proportion of COVID-19 cases in the American population at large, a metric called “prevalence.”* Nobody knows the true number of Americans who have been exposed to or infected with the coronavirus, though attempts to produce much sharper estimates of that figure through blood testing are under way. Prevalence is a crucial number for epidemiologists, in part because it lets them calculate a pathogen’s true infection-fatality rate: the number of people who die after becoming infected.
But the positivity rate is still valuable. “It’s not a normal metric, but it can be a very useful one in some circumstances,” Andrews said. The test-positivity rate is often used to track the spread of rare but deadly diseases, such as malaria, in places where most people aren’t able to get tested, he said. And if the same proportion of a population is being tested over time, the test-positivity rate can even be used to calculate the contagiousness of a disease.
Because the number of Americans tested for COVID-19 has changed over time, the U.S. test-positivity rate can’t yet provide much detailed information about the contagiousness or fatality rate of the disease. But the statistic can still give a rough sense of how bad a particular outbreak is by distinguishing between places undergoing very different sizes of epidemics, Andrews said. A country with a 25 percent positivity rate and one with a 2 percent positivity rate are facing “vastly different epidemics,” he said, and the 2 percent country is better off.
In that light, America’s 20 percent positivity rate is disquieting. The U.S. did almost 25 times as many tests on April 15 as on March 15, yet both the daily positive rate and the overall positive rate went up in that month. If the U.S. were a jar of 330 million jelly beans, then over the course of the outbreak, the health-care system has reached in with a bigger and bigger scoop. But every day, 20 percent of the beans it pulls out are positive for COVID-19. If the outbreak were indeed under control, then we would expect more testing—that is, a larger scoop—to yield a smaller and smaller proportion of positives. So far, that hasn’t happened.
In an ideal testing regime—and in any of the testing regimes that experts say must exist before the United States can end its lockdowns—anyone with a fever and a dry cough would be tested immediately. A very large portion, if not most, of those people would turn out not to be infected with the new coronavirus, SARS-CoV-2, because humans are susceptible to many other respiratory infections. But when tests are rationed so strictly, only people with severe symptoms make it into the testing pool, ensuring that the positivity rate will be extremely high.
Local rationing rules are not the only reason that Americans are not getting tested. Some people live in a place that’s not doing much testing at all, either because doctors’ offices have no tests to offer or because of an already strained or nonexistent local health-care system. Others avoid the doctor if they’re sick, or never get sick enough to seek a test—but if the U.S. were testing more people, as experts say it must, then general-population surveillance or workplace testing could detect their illness, too.
The test-positivity rate, then, is a decent (if unusual) proxy for the severity of an outbreak in an area. And it shows clearly that the U.S. still lags far behind other countries in the course of fighting its outbreak. South Korea—which discovered its first coronavirus case on the same day as the U.S.—has tested more than half a million people, or about 1 percent of its population, and discovered about 10,500 cases. The U.S. has now tested 3.2 million people, which is also about 1 percent of its population, but it has found more than 630,000 cases. So while the U.S. has a 20 percent positivity rate, South Korea’s is only about 2 percent—a full order of magnitude smaller.
South Korea is not alone in bringing its positivity rate down: America’s figure dwarfs that of almost every other developed country. Canada, Germany and Denmark have positivity rates from 6 to 8 percent. Australia and New Zealand have 2 percent positivity rates. Even Italy—which faced one of the world’s most ravaging outbreaks—has a 15 percent rate. It has found nearly 160,000 cases and conducted more than a million tests. Virtually the only wealthy country with a larger positivity rate than the U.S. is the United Kingdom, where more than 30 percent of people tested for the virus have been positive.
Comparing American states to regions in other countries results in the same general pattern. In Lombardy, the hardest hit part of Italy, the positive rate today stands at about 28 percent. That’s comparable to the rate in Connecticut. But New York, so far the hardest hit state in the U.S., has an even higher rate of 41 percent. And in New Jersey, an astounding one in two people tested for the virus are found to have it.
The prevalence of COVID-19 might be higher in the New York area than anywhere else in the country, but high test-positivity rates are not confined to the mid-Atlantic. Five other states have a positive rate above 20 percent: Michigan, Georgia, Massachusetts, Illinois, and Colorado. They are spread across the country, and they all have obviously serious outbreaks. Each of the eight states with positive rates over 20 percent has, individually, reported more COVID-19 deaths than South Korea.
Hawaii, meanwhile, has tested twice as many people per capita as Illinois, but its positivity rate is only one-tenth as high as the larger state’s. As the outbreak comes under control, more states should have positivity rates closer to Hawaii’s, Andrews, the Stanford professor, said. At the beginning of a pandemic, both the actual number of infections and the number of tests per day shoot up, and the positivity rate is controlled by whichever happens to grow faster, he said. In this case, the faster-growing number appears to have been infections. “As things stabilize, if the testing rate declines and the positivity rate declines, you have some good signal that the epidemic is declining,” he said.
Not every epidemiologist feels as comfortable drawing conclusions from the test-positivity rate as Andrews. “If you want to interpret [the positivity rate] as a hint to prevalence in a particular location, you have to assume lots of other things stay constant,” Daniel Westreich, an epidemiology professor at the University of North Carolina, told us. He warned that too little was still known about who exactly is getting tested, and how reliable the tests are, to draw large conclusions from the positivity rate alone.
“We just haven’t tested enough people yet,” he said. “If you were doing random screening of the whole population, we just don’t know what you’d see. We don’t know how many asymptomatic viral shedders are out there.” As such, he advised extreme caution in using the rate—but being cautious about data, he added, “is my job.”
We feel confident reporting the U.S. test-positivity rate now for several reasons. First, we know that when states and cities ration tests, they do so by imposing criteria that allow for only the sickest or the most vulnerable people, such as residents of nursing homes, to get tested. We know that in states with a very high test-positivity rate, such as New Jersey, many people are still dying in nursing homes without getting tested. And we know that, even though a wide variety of nose-swab tests are being used across the country, the type of test used—called a polymerase chain reaction, or PCR, test—is generally very reliable. Westreich and Andrews said that any PCR test was “pretty good” at detecting true negatives.
Finally, the test-positivity rate has become much more reliable nationwide over the past few weeks. As recently as the end of March, not all states reported every negative test result from commercial laboratories. Nearly every state now publishes those numbers.
While our numbers still probably do not capture every coronavirus test in the U.S., outside evidence now suggests that our data are fairly complete. When the White House Coronavirus Task Force has reported the number of tests completed nationwide, its numbers have broadly matched the COVID Tracking Project’s. In addition, the largest commercial-test processors, Quest and LabCorp, have released top-line statistics that align with ours at the COVID Tracking Project.
The high positivity rate also suggests that new cases in the U.S. have plateaued only because the country has hit a ceiling in its testing capacity. Looking solely at positives, the U.S. is steaming toward 650,000 confirmed cases, but the number of new cases per day appears to be plateauing or even declining.

There are several ways to interpret this development. It might suggest, for instance, that the more than 3.2 million tests completed in the U.S. over the past two months have finally captured a good chunk of the people who are actually infected. While it’s clear that the country is not capturing every case, this decline in new positive cases might suggest the country has started to get the virus’s spread under control.
But there is another way to interpret the decline in new cases: The growth in the number of new tests completed per day has also plateaued. Since April 1, the country has tested roughly 145,000 people every day with no steady upward trajectory. The growth in the number of new cases per day, and the growth in the number of new tests per day, are very tightly correlated.
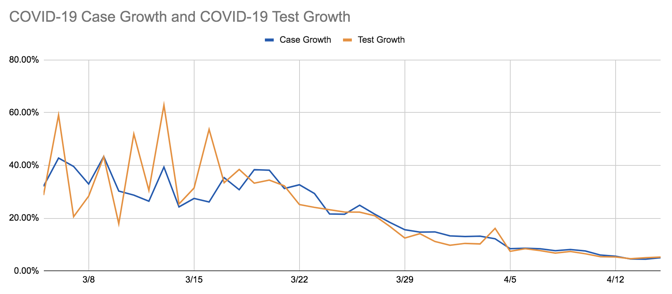
This tight correlation suggests that if the United States were testing more people, we would probably still be seeing an increase in the number of COVID-19 cases. And combined with the high test-positivity rate, it suggests that the reservoir of unknown, uncounted cases of COVID-19 across the country is still very large.
Each of those uncounted cases is a small tragedy and a microcosm of all the ways the U.S. testing infrastructure is still failing. When Sarah Pavis, a 36-year-old engineer in New York, woke up on Tuesday, she was out of breath and her heart was racing. An hour of deep breathing failed to calm her pulse. When her extremities started tingling, she called 911. It was her ninth day of COVID-19 symptoms.
New York City’s positivity rate is an astonishing 55 percent. More than 111,000 of the city’s residents have lab-confirmed cases of COVID-19, but Pavis is not among them. When the ambulance arrived at Pavis’s apartment, an EMS worker took her vitals, then explained there was little he could do to help. The city’s hospitals only admitted people with a blood-oxygen level of 94 percent or lower, he said. Pavis’s blood-oxygen reading was 96 percent. That 2 percent difference meant that her illness was not serious enough to merit hospitalization, not serious enough to be tested, not serious enough to be counted.

Workers say too little is being done to enforce social distancing in stores, and some are not given masks or training on cleaning.
Whole Foods workers across the US are planning to hold another sickout protest on 1 May, as the number of confirmed cases of coronavirus infections at the supermarket chain continues to rise and workers charge the Amazon-owned company is doing too little to help them.
Workers complain too little is being done to enforce social distancing in stores; it is difficult, and sometimes impossible, to qualify for sick pay; and some are not given masks or training on cleaning. In the meantime, Whole Foods is reportedly recording record sales.
Dan Steinbrook, an employee at Whole Foods in Boston, said: “The bottom line is we don’t think Whole Foods or Amazon is doing nearly enough as they could be to protect both employees and customers at the store in terms of personal safety and public health.”
Steinbrook, who also participated in a sickout protest on 31 March organized by Whole Worker, a worker activism group said: “Grocery stores are one of the only places open to the public so they’ve become a significant public health concern in terms of stopping the spread of this disease. Any transmission we can stop at the grocery stores is extremely important for saving a lot of lives.”
Whole Foods workers have become increasingly concerned over the confirmed cases of coronavirus at Whole Foods stores. Employees have tested positive for coronavirus at Whole Foods locations across the country including West Orange, New Jersey; Sudbury, Massachusetts; Brookline, Massachusetts; Arlington, Massachusetts; Hingham, Massachusetts; Cambridge, Massachusetts; San Francisco, California; New York City, New York; Fort Lauderdale, Florida; New Orleans, Louisiana; and Allentown, Pennsylvania.
The Guardian spoke to several Whole Foods workers across the US about working conditions and the company’s policies. The workers requested to remain anonymous for fear of retaliation.
“I haven’t felt safe going into work because Whole Foods hasn’t really done anything to combat the amount of Amazon shoppers in the stores,” said a Whole Foods employee at Bowery Place in New York City, the center of the coronavirus pandemic in the US. “The store has been closing earlier, but they still want us to stay until 11pm to clean, and we aren’t trained to clean or given masks or anything.”
Whole Foods workers have noted some stores where a worker has tested positive for coronavirus have yet to be publicly reported in the media.
“Team members are being told there was a deep clean overnight and not to worry,” said a Whole Foods worker in West Bloomfield, Michigan. “I’m scared to work. I have three immune sensitive people living in my house and I don’t want to get them sick, but I can’t lose my only income.”
A worker at Whole Foods in Chapel Hill, North Carolina, said there have been two positive cases at their store. “It has been almost impossible to maintain basic social distancing practices. We’ve seen huge sales ever since the outbreak and it’s been all hands on deck. As of 1 April, there were no limits on the number of customers allowed in at a given time,” said the employee.
In Minnesota, a Whole Foods employee is currently on unpaid leave after experiencing coronavirus symptoms when their roommate was advised by their doctor to self-quarantine.
“When I talked to my HR department they told me I would need to take a two week leave as well, but unless I test positive for Covid-19, I do not qualify for the ‘guaranteed two weeks paid time off’ corporate is saying they are offering,” said the worker. “Everyone knows tests are limited and unavailable to most people unless they are showing severe symptoms, and as retail workers, many of us cannot afford to go to the doctor unless we’re in desperate need of medical attention.”
A Whole Foods employee in Massachusetts is also currently taking unpaid leave after experiencing coronavirus symptoms.
“I’m in a situation where I can’t get tested or afford a doctor. At first I was told I wouldn’t be eligible for sick pay without a positive test. Later I was told that I might qualify, that pay was being disbursed on a case by case basis. My case has been pending for over a week with no response and I ran out of paid time off,” said the worker.
“My parents lent me money, so I’ll be able to finish quarantine and still afford groceries. Money was tight before bills were due, and those fears kept me from reaching out to a doctor. My symptoms were mild, but I don’t know what I would have done if they got serious.”
A Whole Foods spokesperson told the Guardian: “The safety of our team members and customers is our top priority and we are diligently following all guidance from local health and food safety authorities. We’ve been working closely with our store Team Members, and are supporting the diagnosed Team Members, who are in quarantine.
“Out of an abundance of caution, each of these stores performed an additional deep cleaning and disinfection, on top of our current enhanced sanitation measures. As we prioritize the health and safety of our customers and Team Members, we will continue to do the following to help contain the spread of Covid-19.”
Early peek at data on Gilead coronavirus drug suggests patients are responding to treatment

A Chicago hospital treating severe Covid-19 patients with Gilead Sciences’ antiviral medicine remdesivir in a closely watched clinical trial is seeing rapid recoveries in fever and respiratory symptoms, with nearly all patients discharged in less than a week, STAT has learned.
Remdesivir was one of the first medicines identified as having the potential to impact SARS-CoV-2, the novel coronavirus that causes Covid-19, in lab tests. The entire world has been waiting for results from Gilead’s clinical trials, and positive results would likely lead to fast approvals by the Food and Drug Administration and other regulatory agencies. If safe and effective, it could become the first approved treatment against the disease.
The University of Chicago Medicine recruited 125 people with Covid-19 into Gilead’s two Phase 3 clinical trials. Of those people, 113 had severe disease. All the patients have been treated with daily infusions of remdesivir.
“The best news is that most of our patients have already been discharged, which is great. We’ve only had two patients perish,” said Kathleen Mullane, the University of Chicago infectious disease specialist overseeing the remdesivir studies for the hospital.
Her comments were made this week during a video discussion about the trial results with other University of Chicago faculty members. The discussion was recorded and STAT obtained a copy of the video.
The outcomes offer only a snapshot of remdesivir’s effectiveness. The same trials are being run concurrently at other institutions, and it’s impossible to determine the full study results with any certainty. Still, no other clinical data from the Gilead studies have been released to date, and excitement is high. Last month, President Trump touted the potential for remdesivir — as he has for many still-unproven treatments — and said it “seems to have a very good result.”
In a statement Thursday, Gilead said: “What we can say at this stage is that we look forward to data from ongoing studies becoming available.”
Gilead had said to expect results for its trial involving severe cases in April. Mullane said during her presentation that data for the first 400 patients in the study would be “locked” by Gilead Thursday, meaning that results could come any day.
Mullane, while encouraged by the University of Chicago data, made clear her own hesitancy about drawing too many conclusions.
“It’s always hard,” she said, because the severe trial doesn’t include a placebo group for comparison. “But certainly when we start [the] drug, we see fever curves falling,” she said. “Fever is now not a requirement for people to go on trial, we do see when patients do come in with high fevers, they do [reduce] quite quickly. We have seen people come off ventilators a day after starting therapy. So, in that realm, overall our patients have done very well.”
She added: “Most of our patients are severe and most of them are leaving at six days, so that tells us duration of therapy doesn’t have to be 10 days. We have very few that went out to 10 days, maybe three,” she said.
Reached by STAT, Mullane confirmed the authenticity of the footage but declined to comment further. In a statement, the University of Chicago Medicine said “drawing any conclusions at this point is premature and scientifically unsound.”
Asked about the data, Eric Topol, director of the Scripps Research Translational Institute, described them as “encouraging.”
“The severely hit patients are at such high-risk of fatality. So if it’s true that many of the 113 patients were in this category and were discharged, it’s another positive signal that the drug has efficacy,” he said, adding that it will be important to see more data from randomized controlled studies.
Gilead’s severe Covid-19 study includes 2,400 participants from 152 different clinical trial sites all over the world. Its moderate Covid-19 study includes 1,600 patients in 169 different centers, also all over the world.
The trial is investigating five- and 10-day treatment courses of remdesivir. The primary goal is a statistical comparison of patient improvement between the two treatment arms. Improvement is measured using a seven-point numerical scale that encompasses death (at worst) and discharge from hospital (best outcome), with various degrees of supplemental oxygen and intubation in between.
The lack of a control arm in the study could make interpreting the results more challenging.
A lack of data has led to yo-yoing expectations for the drug. Two studies in China had enrollment suspended partway through because there were not enough patients available. A recent report of patients given the drug under a special program to make it available to those who are very ill generated both excitement and skepticism.
In scientific terms, all the data are anecdotal until the full trial reads out, meaning that they should not be used to draw final conclusions. But some of the anecdotes are dramatic.
Slawomir Michalak, a 57-year-old factory worker from a suburb west of Chicago, was among the participants in the Chicago study. One of his daughters started feeling ill in late March and was later diagnosed with mild Covid-19. Michalak, by contrast, came down with a high fever and reported shortness of breath and severe pain in his back.
“It felt like someone was punching me in the lungs,” he told STAT.
At his wife’s urging, Michalak went to the University of Chicago Medicine hospital on Friday, April 3. His fever had spiked to 104 and he was struggling to breath. At the hospital, he was given supplemental oxygen. He also agreed to participate in Gilead’s severe Covid-19 clinical trial.
His first infusion of remdesivir was on Saturday, April 4. “My fever dropped almost immediately and I started to feel better,” he said.
By his second dose on Sunday, Michalak said he was being weaned off oxygen. He received two more daily infusions of remdesivir and recovered enough to be discharged from the hospital on Tuesday, April 7.
“Remdesivir was a miracle,” he said.
The world is waiting to find out if it is really so.