
Category Archives: Hope
Cartoon – State of the Union (Office Visit)
Window Cleaners at Children’s Hospitals







Cartoon – The Great Ledger of Life

Cartoon – 2021 New Year

This terrible year taught me something about hope

The first month of the pandemic was also supposed to be the month I got pregnant, but my clinic closed and plans changed. Doctors and nurses needed personal protective equipment to tend to patients with covid-19, not women with recurrent miscarriages.
When the clinic reopened several months later, it turned out my husband and I had only been delaying yet another loss: In late August, he obeyed the medical center’s strict coronavirus protocols by waiting anxiously in the car while I trudged inside, masked and hand-sanitized, to receive a miscarriage diagnosis alone. I searched the ultrasound screen for the rhythmic beat of a heart, and then accepted that whatever had once been there was now gone.
But that was 2020 for you, consistent only in its utter crappiness. For every inspiring video of neighbors applauding a shift change at the hospital, another video of a bone-tired nurse begging viewers to believe covid was real, it wasn’t a hoax, wear a mask.
For every protest organized by activists who understood racism is also a long-term crisis, an appearance by the Proud Boys; for every GoFundMe successfully raising money for a beloved teacher’s hospital bills, a bitter acknowledgment that online panhandling is our country’s version of a safety net.
Millions of citizens stood in line for hours to vote for the next president and then endured weeks of legal petitions arguing that their votes should be negated. The basis for these legal actions were conspiracy theories too wild to be believed, except that millions of other citizens believed them.
And that was 2020 for you, too: accepting the increasingly obvious reality that the country was in peril, built on iffy foundations that now buckled under pressure. My loved ones who worked as waiters or bartenders or physical therapists were choosing between health and paychecks, and even from the lucky safety of my work-from-home job, each day began to feel like watching America itself arrive at a hospital in bad shape, praying that doctors or clergy could find something they were able to save.
Is there a heartbeat?
You want the answer to be yes, but even so, it was hard to imagine how we would come back from this.
What kind of delusional person would even try to get pregnant in this world? In my case it would never be a happy accident; it would always be a herculean effort. And so it seemed I should have some answers.
How do you explain to a future child: Sorry, we can’t fix climate change; we can’t even get people to agree that we should wear masks in grocery stores? How do you explain the frustration of seeing brokenness, and then the wearying choice of trying to fix it instead of abandoning it? How do you say, Love it anyway. You’re inheriting an absolute mess, but love it anyway?
I found myself asking a lot of things like this in 2020, but really they were all variations of the same question: What does it mean to have hope?
But in the middle of this, scientists worked quietly in labs all over the world. They applied the scientific method with extraordinary discipline and speed. A vaccine was developed. Tens of thousands of volunteers rolled up their sleeves and said, Try it out on me.
It was approved, and a nurse from Long Island was the first American televised receiving it. Her name was Sandra Lindsay, an immigrant from Jamaica who had come to the United States 30 years ago and who had spent the last year overseeing critical care teams in back-to-back shifts. She said she had agreed to go first to show communities of color, long abused, brushed-off or condescended to by the medical system, that the vaccine was safe.
Here was hope. And more than that, here was hope from a woman who had more reason than most to be embittered: an exhausted health-care worker who knew too well America’s hideous racial past and present, who nonetheless also knew there was only one way out of the tunnel. Here she was, rolling up her own sleeve, and there were the lines of hospital employees ready to go after her, and there were the truck drivers ferrying shipments of syringes.
I can’t have been the only person to watch the video of those early inoculations, feeling elated and tired, and to then burst into tears. I can’t have been the only person to realize that even as 2020 revealed brokenness, it also contained such astounding undercurrents of good.
The scientific method works whether you accept it or not. Doctors try to save you whether you respected public-health guidelines or not. Voter turnout was astronomical because individual citizens realized they were all, every one of them, necessary pieces in a puzzle, even if they couldn’t see what the final picture was supposed to look like.
The way to believe in America is to believe those things are passed down, too.
Sometime in October, a couple of months after my last miscarriage — when the country was riding up on eight months of lonely and stoic birthdays, graduations, deaths and weddings — I went into the bathroom and saw a faint second line on a First Response pregnancy test. It was far from my first rodeo, so I knew better than to get excited. I mentioned it to my husband with studied nonchalance, I told him that I’d test again in a few days but that we should assume the worst would happen.
Two weeks after that, I had a doctor’s appointment, and then another a week later, each time assuming the worst, but each time scheduling another appointment anyway, until eventually I was further along than I’d ever gotten before — by one day, then three days, then thirty.
I am not a superstitious person. I don’t believe that good things always come to those who deserve them. I believe that stories regularly have sad endings and that it’s often nobody’s fault when they do, and that we should tell more stories with sad endings so that people who experience them know that they’re not alone.
But 2020 has taught me that I am, for better or worse, someone who wants to hope for things. To believe in the people who developed vaccines. In the people who administered them. In Sandra Lindsay. In the people who delivered groceries, who sewed masks, who have long cursed America’s imperfect systems and long fought to change them, who still donate $10 to a sick teacher’s GoFundMe.
At my most recent appointment, the doctor’s office was backed up in a holiday logjam. I sat in the exam room for nearly three hours while my husband again waited anxiously in the car. I texted him sporadic updates and tried to put hope in a process that so far had not seemed to warrant my hope.
It all felt precarious. The current reality always feels precarious.
And yet there we all are together, searching for signs of life, hoping that whatever we emerge to can be better than what we had before, and that whatever we build will become our new legacy. The sonographer finally arrived and turned on the machine.
There was a heartbeat. There was a heartbeat.
America hopes 2021 will be less terrible
https://www.axios.com/axios-surveymonkey-poll-2021-hopes-7de84661-c2dd-43df-9509-3dda4a35a9f5.html
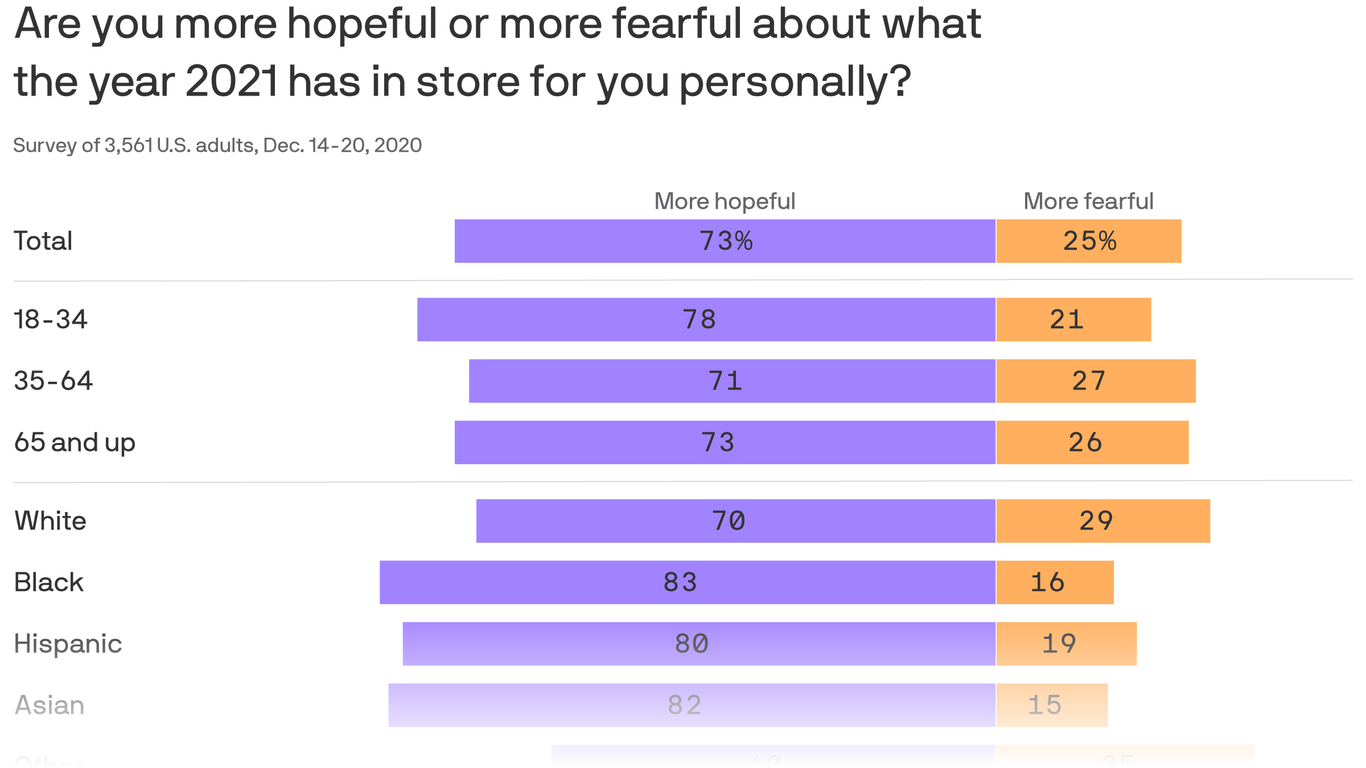
More than six out of 10 Americans are hopeful about what 2021 has in store for the world, according to a new Axios/SurveyMonkey poll.
The big picture: After a year dominated by the pandemic and a seemingly endless presidential election, Americans are overwhelmingly hopeful that things will get better with the pandemic — and more narrowly hopeful about Joe Biden’s presidency.
By the numbers: 63% of poll respondents said they’re more hopeful than fearful about what 2021 holds in store for the world, while 36% said they’re more fearful.
- That’s a jump in optimism compared to the same poll heading into 2019, when just 51% said they were hopeful and 48% said they were fearful.
- The only group that wasn’t optimistic about 2021 was Republicans: 41% said they were more hopeful, while 58% said they were more fearful.
Between the lines: Americans were even more optimistic about the year ahead for them personally — mostly driven by the hopes of young adults, people of color and Democrats.
The coronavirus was the one issue that united most people in optimism. Overall, 76% were more hopeful than fearful about the pandemic next year — a view that held across most age groups, racial and ethnic groups, and parties.
- 82% of Democrats, 72% of Republicans, and 73% of independents said they were more hopeful than fearful.
The Biden presidency was more divisive. Overall, 56% were more hopeful about his presidency, while 42% were more fearful.
- Not surprisingly, Republicans are the most pessimistic: 82% said they’re more fearful than hopeful about his presidency.
- By contrast, 59% of independents said they’re more hopeful about it — and 92% of Democrats said the same.
And while Republicans are ready for President Trump to take on a big leadership role in the Republican Party after his presidency ends, that’s not true of everyone else.
- 75% of Republicans said they’re more concerned that Trump will play too small a role in the future of the GOP rather than too big a role — while 51% of all respondents said they’re more concerned that he’ll play too big a role.
- And more than half of Republicans (52%) said they believe Trump will have a major role in the Republican Party, while 30% said they think he’ll have a minor role.
- By contrast, just 34% of all respondents expect him to play a major role, while 32% think he’ll have a minor role and 31% think he’ll have no role at all.
Cartoon – Hope for the New Year
Major study finds common steroid reduces deaths among patients with severe Covid-19
Major study finds common steroid reduces deaths among patients with severe Covid-19

A cheap, readily available steroid drug reduced deaths by a third in patients hospitalized with Covid-19 in a large study, the first time a therapy has been shown to possibly improve the odds of survival with the condition in the sickest patients.
Full data from the study have not been published or subjected to scientific scrutiny. But outside experts on Tuesday immediately embraced the top-line results. The drug, dexamethasone, is widely available and is used to treat conditions including rheumatoid arthritis, asthma, and some cancers.
In a statement, Patrick Vallance, the U.K. government’s chief scientific adviser, called the result “tremendous news” and “a ground-breaking development in our fight against the disease.” Scott Gottlieb, a former commissioner of the U.S. Food and Drug Administration, called it “a very positive finding” in an interview on CNBC. “I think it needs to be validated, but it certainly suggests that this could be beneficial in this setting.”
Atul Gawande, the surgeon, writer and public health researcher, urged caution, tweeting, “after all the retractions and walk backs, it is unacceptable to tout study results by press release without releasing the paper.”
The study randomly assigned 2,104 patients to receive six milligrams of dexamethasone once a day, by mouth or intravenous injection. These were compared to 4,321 patients assigned to receive usual care alone.
In patients who needed to be on a ventilator, dexamethasone reduced the death rate by 35%, meaning that doctors would prevent one death by treating eight ventilated patients. In those who needed oxygen but were not ventilated, the death rate was reduced 20%, meaning doctors would need to treat 25 patients to save one life. Both results were statistically significant.
There was no benefit in patients who didn’t require any oxygen. The researchers running the study, called RECOVERY, decided to stop enrolling patients on dexamethasone on June 8 because they believed they had enough data to get a clear result.
“Dexamethasone is the first drug to be shown to improve survival in COVID-19,” Peter Horby, one of the lead investigators of the study and a professor in the Nuffield Department of Medicine at the University of Oxford, said in a statement. He added that the drug should now become the standard treatment for patients with Covid-19 who need oxygen. “Dexamethasone is inexpensive, on the shelf, and can be used immediately to save lives worldwide.’”
A different arm of the same study showed on June 5 that hydroxychloroquine, widely touted as a potential Covid treatment, had no benefit in hospitalized patients. Yesterday, based in part on those results, the Food and Drug Administration revoked an Emergency Use Authorization for using hydroxychloroquine in those patients.
From the start of the pandemic in March, researchers have focused on two different stages of Covid-19, which will likely require very different interventions. Some drugs are designed to directly combat the novel coronavirus, SARS-CoV-2, that causes the disease. The first medicine shown to have a benefit, remdesivir from the biotech firm Gilead Sciences, falls into this category, even though, because it must be given intravenously, it has been tested in hospitalized patients. Remdesivir shortens the course of infection, but has not been shown to save lives.
After patients have become profoundly sick, the problem starts to become not only the virus but their own immune system, which attacks the lungs, a condition called acute respiratory distress syndrome, or ARDS. For these patients, doctors have believed, they would need to dampen patients’ immune response even as they fought the virus.
Initially, excitement in this area fell on new and expensive drugs, such as Actemra, a rheumatoid arthritis drug from Roche that is used to treat a similar condition caused by some cancer immunotherapies. But a study in patients who needed oxygen showed no benefit from a similar drug, although another arm in sicker patients is continuing. The National Institutes of Health is conducting a study of an Eli Lilly pill targeting rheumatoid arthritis, an extension of the study that showed remdesivir has a benefit.
Dexamethasone, which reached the market 59 years ago, seemed an unlikely candidate to help these patients; it was seen as too crude a way of tamping down the immune system. In guidelines for physicians treating the disease, the NIH doesn’t even mention the therapy.
Studies that are testing other medicines may now need to incorporate the use of the drug, which could complicate analyzing the results. A spokesperson for Regeneron, which is testing Covid-19 drugs focused on both attacking the virus and dampening the immune system, said the company’s studies are written so that when a new medicine becomes the standard of care, it becomes available to patients in the trial.
Some studies have shown a benefit for using dexamethasone in acute respiratory distress syndrome not related to Covid-19, although the benefit was smaller than in RECOVERY.
The result, should it hold up to further scrutiny, shows the benefit of the strategy of Horby and Martin Landray, the Oxford researchers who designed the study, leveraging the U.K. health system to start a study of multiple inexpensive potential Covid-19 therapies — including hydroxychloroquine, dexamethasone, and also some older HIV medicines. Several months into the Covid-19 pandemic, two of the most important results come from this single study.
Neither of those results, however, have been scrutinized or published.
Coronavirus: 15 emerging themes for boards and executive teams

Board directors and executives can pool their wisdom to help companies grapple with the challenge of a lifetime.
As Winston Churchill said, “Now this is not the end. It is not even the beginning of the end. But it is, perhaps, the end of the beginning.” We are seeing some faint signs of progress in the struggle to contain the pandemic. But the risk of resurgence is real, and if the virus does prove to be seasonal, the effect will probably be muted. It is likely never more important than now for boards of directors and executive management teams to tackle the right questions and jointly guide their organizations toward the next normal.
Recently, we spoke with a group of leading nonexecutive chairs and directors at companies around the world who serve on the McKinsey Resilience Advisory Council. They generously shared the personal insights and experiences gained from their organizations’ efforts to manage through the crisis and resume work. The 15 themes that emerged offer a guide to boards and executive teams everywhere. Together, they can debate these issues and set an effective context for the difficult decisions now coming up as companies plan their return to full activity.
Managing through the crisis
1. Boards must strike the right balance between hope for the future and the realism that organizations need to hear. There are many prognostications on what comes after COVID-19. Many will be helpful. Some will be right. Boards and managers may have some hopes and dreams of their own. Creating value and finding pockets of growth are possible. It is important to have these aspirations, because they form the core of an inner optimism and confidence that organizations need. However, leaders should not conflate aspirations with a prescience about the future.
2. The unknown portion of the crisis may be beyond anything we’ve seen in our professional lives. Boards and managers feel like they might be grappling with only 5 percent of the issues, while the vast majority are still lurking, unknown. Executives are incredibly busy, fighting fires in cash management and other areas. But boards need to add to their burden and ask them to prepare for a “next normal” strategy discussion. Managers need to do their best to find out what these issues are, and then work with boards to ensure that the organization can navigate them. The point isn’t to have a better answer. The point is to build the organizational capability to learn quickly why your answer is wrong, and pivot faster than your peers do. Resilience comes through speed. This may be a new capability that very few organizations have now, and they will likely need to spend real time building it.
3. Beware of a gulf between executives and the rank and file. Top managers are easily adapting to working from home and to flexible, ill-defined processes and ways of working, and they see it as being very effective and also the wave of the future. Many people in the trenches think it is the worst thing to happen to them (even those that are used to working remotely). Remote working is raising the divide between elites and the common man and woman. There is a real risk of serious tension in the social fabric of organizations and in local and national communities.
4. Don’t overlook the risks faced by self-employed professionals, informal workers, and small businesses. These groups are often not receiving sufficient support. But their role in the economy is vital, and they may be noticed only later, when it is too late.
5. Certain industries and sectors are truly struggling and require support. Several disrupted industries and many organizations in higher education, the arts, and sports are severely struggling and require support to safeguard their survival.
Return to work—the path ahead
6. Mid- to long-term implications and scenarios vary considerably. It’s important to differentiate between industries and regions. Some industries may never come back to pre-COVID-19 levels.
7. What went wrong? Boards and executives, but also academics, need to debate the question. Where should we have been focusing? Take three examples. Why did companies ignore the issue of inadequate resilience in their supply chain? The risks of single sourcing were well known and transparent. Also, why did we move headlong toward greater specialization in the workforce, when we knew that no single skill was permanently valuable? Finally, why did we refuse to evolve our business models, although we knew that technology and shifts in societal preferences were forcing us down a treadmill of ever decreasing value-creation potential?
8. How can we prevent a backlash to globalization? The tendency toward nationalism was already strong and is growing during the crisis. The ramifications will be challenging. For example, in pharmaceutical development, residents of the country where a pharma company has its headquarters may expect to get the drug first. Global companies, despite their experience, may find it harder to address and engage directly with diverse, volatile, and potentially conflicting stakeholders. In such times, societies may need someone to mediate between the private sector and some of these stakeholders.
9. Companies need help with government relations. Strong government interventions are occurring on the back of a serious loss of confidence in free-market mechanisms. There is little question that different governments will land on different answers to the debate around how free markets really ought to be structured. The corporate community has been thrust into a new relationship with government, and it is struggling. The government landscape is fragmented, with highly varied approaches and competencies. Companies are looking for a playbook; no one has an infrastructure to manage this complexity.
10. Where will the equity come from, and with what strings attached? Governments are propping up various sectors with new capital. What will they receive in return? Will they distort markets? How can companies manage this process carefully to emerge from the crisis with a stronger balance sheet? Further, much more capital is likely needed; presumably some of it will come from the private sector. Will capital markets be effective and trusted in such times? Who governs this overall process, and what role should the government play? Is it the time for more state funds?
11. The balance between profits and cash flow is tricky, and essential to get right. Many companies are caught right now and are sacrificing their bottom line in order to pay for their financing. That’s not sustainable; companies will need guidance on how to balance the two.
12. It may be time for responsible acquisitions, including to help restructure certain industries. Many “resilients” have “kept their powder dry,” and are now ready to acquire. But they need to be sensitive and allow sellers a good path to exit. We need guidelines for responsible acquisitions.
13. Cyberrisk is growing. Remote working increases the “attack surface” for criminals and state actors. Both are more active. Chief information officers and chief information security officers are grappling with the overwhelming demand for work-from-home technology and the need for stringent cybersecurity.
14. Innovation may never have been so important. Innovation has always been essential to solving big problems. The world is looking not just for new things but also for new ways of doing things (especially on the people side, where we need new behaviors, long-term rather than short-term), capabilities, and work ethics.
15. The path ahead will surely have ups and downs and will require resilience. As lockdowns are relaxed, and segments of the economy reopen, viral resurgences and unforeseen events will keep growth from being a straight line going up. It will likely be a lengthy process of preserving “lives and livelihoods” over several months, if not years. The reality is that many or even most business leaders made choices over the past decades that traded resilience for a perceived increase in shareholder value. Now may be the moment to consider that the era of chipping away at organizational resilience in the name of greater efficiency may have reached its limits. This is not to say that there are no efficiencies to be sought or found, but more that the trade-off between efficiency and resiliency needs to be defined far more clearly than it has been in recent years.
It is the board’s responsibility to coach and advise its management team, especially when the terrain is trickier than usual. However, boards should not mistake the need for vigorous debate with the need for consensus. More than ever, a bias to action is essential, which will frequently mean getting comfortable with disagreement. Apart from all the operational focus needed for the return to work, it is even more important that boards and management teams take a step back to reflect upon these 15 core themes. In summary:
- Take the time to recognize how the people who (directly or indirectly) depend on the company feel.
- Have aspirations about the post-COVID world, but build the resilience to make them a reality.
- Strengthen your capability to engage and work with regulators and the government.
- Watch out for non-COVID risks, and make sure to carve out time to dedicate to familiar risks that have never gone away.
- Find out what went wrong, and answer the uncomfortable truths that investigation uncovers.



