
Cartoon – The Right Vaccine



As President Trump pushes the possibility of a vaccine this year, the C.D.C. has outlined technical scenarios to state public health officials for an unidentified Vaccine A and Vaccine B.
The Centers for Disease Control and Prevention has notified public health officials in all 50 states and five large cities to prepare to distribute a coronavirus vaccine to health care workers and other high-risk groups as soon as late October or early November.
The new C.D.C. guidance is the latest sign of an accelerating race for a vaccine to ease a pandemic that has killed more than 184,000 Americans. The documents were sent out on the same day that President Trump told the nation in his speech to the Republican National Convention that a vaccine might arrive before the end of the year.
Over the past week, both Dr. Anthony S. Fauci, the country’s top infectious disease expert, and Dr. Stephen Hahn, who heads the Food and Drug Administration, have said in interviews with news organizations that a vaccine may be available for certain groups before clinical trials have been completed, if the data is overwhelmingly positive.
Public health experts agree that agencies at all levels of government should urgently prepare for what will eventually be a vast, complex effort to vaccinate hundreds of millions of Americans. But the possibility of a rollout in late October or early November has heightened concerns that the Trump administration is seeking to rush the distribution of a vaccine — or simply to hype that one is possible — before Election Day on Nov. 3.
For an administration that has struggled with the logistical challenges of containing the coronavirus, the distribution of millions of vaccines that must be stored in subzero temperatures and provided first to high-risk groups through America’s flawed, fragmented health care system would be a daunting challenge. Even the C.D.C.’s guidance acknowledged that its plan was hypothetical and based on the need to immediately begin organizing the gigantic effort that would be required if the F.D.A. were to allow the use of a vaccine or two this year.
The C.D.C. plans lay out technical specifications for two candidates described as Vaccine A and Vaccine B, including requirements for shipping, mixing, storage and administration. The details seem to match the products developed by Pfizer and Moderna, which are the furthest along in late-stage clinical trials. On Aug. 20, Pfizer said it was “on track” for seeking government review “as early as October 2020.”

Credit…
“This timeline of the initial deployment at the end of October is deeply worrisome for the politicization of public health and the potential safety ramifications,” said Saskia Popescu, an infection prevention epidemiologist based in Arizona. “It’s hard not to see this as a push for a pre-election vaccine.”
Three documents were sent to public health officials in all states and territories as well as officials in New York, Chicago, Philadelphia, Houston and San Antonio on Aug. 27. They outlined detailed scenarios for distributing two unidentified vaccine candidates, each requiring two doses a few weeks apart, at hospitals, mobile clinics and other facilities offering easy access to the first targeted recipients.
The guidance noted that health care professionals, including long-term care employees, would be among the first to receive the product, along with other essential workers and national security employees. People 65 or older, as well as Native Americans and those who are from “racial and ethnic minority populations” or incarcerated — all communities known to be at greater risk of contracting the virus and experiencing severe disease — were also prioritized in the documents.
That’s a positive development, “so it doesn’t just all wind up in high-income, affluent suburbs,” said Dr. Cedric Dark, an emergency medicine physician at Baylor College of Medicine in Texas.
The C.D.C. noted in its guidance that “limited Covid-19 vaccine doses may be available by early November 2020.” The documents were dispatched the same day that Dr. Robert Redfield, director of the C.D.C., sent a letter to governors asking them to prepare vaccine distribution sites by Nov. 1, as McClatchy reported.
The agency also said its plans were as yet hypothetical, noting, “The Covid-19 vaccine landscape is evolving and uncertain, and these scenarios may evolve as more information is available.” A C.D.C. spokeswoman confirmed that the documents were sent but declined to comment further.
Many of the details listed for the two vaccines — including required storage temperature, the number of days needed between doses, and the type of medical center that can accommodate the product’s storage — match what Pfizer and Moderna have said about their products, which are based on so-called mRNA technology. Neither company responded to requests for comment.
The scenarios, which assume that the two vaccines will demonstrate sufficient safety and effectiveness for an emergency authorization from the F.D.A. by the end of October, noted that Vaccine A, which seems to match Pfizer’s, would have about two million doses ready within this time frame, and that Vaccine B, whose description matches Moderna’s, would have about one million doses ready, with tens of millions of doses of each vaccine ready by the end of the year. Although it’s possible that some promising preliminary data may emerge by the end of October, experts are skeptical.
“The timeline that’s reported seems a bit ambitious to me,” Dr. Dark said. “October’s like 30 days away.”
Trials that test a vaccine’s effectiveness can take years to yield reliable results. It’s possible to draw conclusions sooner “if there is an overwhelming effect” in which vaccinated people appear to be far better protected from disease, said Padmini Pillai, a vaccine researcher and immunologist at M.I.T.
But there can be significant risks in approving a vaccine for broad use in the public before Phase 3 clinical trials involving tens of thousand of participants are completed. Rare but dangerous side effects may only surface over time, after such large numbers of people have received the vaccine.
And data gathered early in a trial might not hold true months down the line. Researchers also need time to test large numbers of people from a variety of backgrounds to determine how well the vaccine works in different populations — including the vulnerable communities identified in the guidelines.
Should any of these snags occur, Dr. Pillai said, “all of this together could diminish public trust in the vaccine.”
James S. Blumenstock, senior vice president of pandemic response and recovery at the Association of State and Territorial Health Officials, confirmed that the three C.D.C. documents were sent to all state and territorial health departments last week. “It is now the time to enhance organizational structure and involve all partners in this planning process going forward,” he said.
Lisa Stromme, a spokeswoman for the Washington State Department of Health, said that her state’s health officials were still at “a very early stage in a planning process,” but were already working toward developing infrastructure that would accommodate the assumptions laid out by the C.D.C.
The C.D.C. documents said that public health administrators should review lessons learned from the 2009 H1N1 pandemic vaccination campaign, which did not have enough doses at the beginning to meet demand.
“It’s good to have a plan out for hospitals and health care systems to prepare” for a potential rollout, said Dr. Taison Bell, a pulmonary and critical care physician at the University of Virginia. But Dr. Bell added that he was concerned that the timeline outlined in the documents “is incredibly ambitious and makes me worry that the administration will prioritize this arbitrary deadline rather than maintaining diligence with following the science.”
The technical comparison of Vaccine A and Vaccine B has some echoes of what was discussed at an Aug. 26 meeting of the Advisory Committee on Immunization Practices of the C.D.C. At the meeting, Dr. Kathleen Dooling, a C.D.C. medical officer, laid out three scenarios: Vaccine A, or the Pfizer vaccine, is approved, Vaccine B, the Moderna vaccine, is approved, or both. The requirement that Pfizer’s vaccine be stored at minus 70 degrees Celsius would mean that it couldn’t be administered at most small sites, she said. The C.D.C. documents noted that orders of Vaccine A would go “to large administration sites only.” The Moderna vaccine requires storage at minus 20 degrees Celsius.
The C.D.C. documents said the vaccine would be free to patients, but that providers might not be reimbursed for administrative costs if the vaccine was given an emergency authorization, rather than a standard approval.
Experts worry that the process is unlikely to go off without a hitch, given the last-minute scramble and the mixed messaging so far. “I think distribution is going to be very tricky for the vaccine, particularly if there is a cold storage requirement,” Dr. Bell said.
There are also likely to be challenges administering both doses of the proposed vaccines, which must be given weeks apart, Dr. Dark said. “How are you going to make sure people get both?”
A Covid-19 vaccine, amazingly, is close. Why am I so worried?

A mere six months after identifying the SARS-CoV-2 virus as the cause of Covid-19, scientists are on the precipice of a having a vaccine to fight it. Moderna and the National Institutes of Health recently announced the start of a Phase 3 clinical trial, joining several others in a constructive rivalry that could save millions of lives.
It’s a truly impressive a feat and a testament to the power of basic and applied medical sciences. Under normal circumstances, vaccine approvals are measured in decades. Milestones that once took months or years have been achieved in days or weeks. If these efforts are successful, the Covid-19 vaccine could take a place alongside the Apollo missions as one of history’s greatest scientific achievements.
I’m optimistic. And yet, as someone who studies drug development, I want to temper expectations with a dose of realism and perhaps a bit of angst. Behind the proud declarations, many science and medical professionals have been whispering concerns. These whispers have escalated into a murmur. It’s time to cry them loudly:
Hey, Food and Drug Administration: Don’t be rash! Premature approval of a sub-standard Covid-19 vaccine could have dire implications, and not just for this pandemic. It could harm public health for years, if not generations, to come.
Unfortunately, elements now in place make such a disastrous outcome not only possible but in fact quite likely. Specifically, the FDA and its staff of chronically overworked and underappreciated regulators will face enormous public and political pressure to approve a vaccine. Whether or not one worries about an “October surprise” aimed at the upcoming election, regulators will be pressed hard. Some will stand firm. Some may resign in protest. But others could break and allow a bad vaccine to be released.
What makes a “bad vaccine”? Insufficient protection against the disease it is designed for, unwanted side effects, or some combination of the two. If an approved Covid-19 vaccine turns out to be ineffective, this could unintentionally promote wider spread of the disease by individuals who presume they were protected from it. Likewise, a negative experience with one vaccine might discourage the use of other vaccines that are far more safe and effective, whether they are for Covid-19 or other vaccine-preventable diseases.
Some things take time. Under normal circumstances, ensuring that a vaccine’s effects are safe and durable requires years of study and monitoring. And there is some evidence that natural immune responses to SARS-CoV-2 infection could be transient, making sustained investigation all the more necessary. A merely short-term effect could encourage vaccinated individuals to resume risky behaviors, which would all but guarantee that the epidemic endures. And if unintended side effects turn out to include, for instance, chronic inflammatory or autoimmune disease, a bad vaccine could impart lifelong damage.
But wait, there’s worse! A bad Covid-19 vaccine could further undermine confidence in the many safe, reliable vaccines already in our public health arsenal. Vaccine skepticism and anti-science bias, propagated by B-list celebrities and Russian troll farms, have been gaining strength all year. Combined with disappointing Covid-19 outcomes, such malign forces could facilitate the reemergence of once-vanquished foes — polio, measles, mumps, rubella, diphtheria, whooping cough, and tetanus — that once killed multitudes of children each year.
These are enormous risks. Placing all of our bets on a small set of untried vaccine technologies would be gobsmackingly foolish. Yet this is exactly what we are now doing. Most of the high-profile names capturing headlines are pursuing comparatively minor variations on a theme of genetic vaccines (those delivered via DNA or RNA). If one approach happens to work, the odds are higher the others will work as well. Disappointing results from one candidate, though, might presage failure across the board.
Rather than investing in a balanced portfolio of vaccines with different approaches — not to mention different therapies, devices, and diagnostics for treating Covid-19 — too many observers, too many companies, and too many governmental officials seem to be narrowly focused on hopes for a “savior” vaccine. Were that savior to fail, our national morale, already low, could plummet even further.
Don’t get me wrong. I, along with millions of Americans, want a Covid-19 vaccine. But we deserve one that’s been proven to be safe and effective.
It’s not too late to take a deep breath and devise a strategy to balance short- and long-term goals, including vaccination, improved diagnostics, and existing and novel treatments. We must support the FDA and hope that its scientists and physicians retain the strength and conviction to resist approving a substandard vaccine.
For encouragement, we should look to Frances Oldham Kelsey, a veritable patron saint of the FDA. In 1960, during her first month working for the agency, Kelsey was asked to approve a sedative called Kevadon, which had the potential to generate billions in revenue. Despite enormous pressure, Kelsey spotted a risk for toxicity and dug in her heels. She refused to rubber stamp the approval. Her actions saved the lives of countless babies. Kevadon, better known as thalidomide, proved to be one of the most dangerous and disfiguring drugs in history.
Kelsey passed away in 2015 at the age of 101. We must pray that her spirit inspires a new generation of FDA leaders with the courage to say, “No.”

The plan, which is co-led by the WHO, the Coalition for Epidemic Preparedness Innovations and Gavi, the vaccine alliance, was of interest to some members of the Trump administration and is backed by traditional U.S. allies, including Japan, Germany and the European Commission, the executive arm of the European Union.
But the United States will not participate, in part because the White House does not want to work with the WHO, which President Trump has criticized over what he characterized as its “China-centric” response to the pandemic.
“The United States will continue to engage our international partners to ensure we defeat this virus, but we will not be constrained by multilateral organizations influenced by the corrupt World Health Organization and China,” said Judd Deere, a spokesman for the White House.
The Covax decision, which has not been previously reported, is effectively a doubling down by the administration on its bet that the United States will win the vaccine race. It eliminates the chance to secure doses from a pool of promising vaccine candidates — a potentially risky strategy.
“America is taking a huge gamble by taking a go-it-alone strategy,” said Lawrence Gostin, a professor of global health law at Georgetown University.
Kendall Hoyt, an assistant professor at Dartmouth’s Geisel School of Medicine, said it was akin to opting out of an insurance policy.
The United States could be pursuing bilateral deals with drug companies and simultaneously participating in Covax, she said, increasing its odds of getting some doses of the first safe vaccine. “Just from a simple risk management perspective, this [Covax decision] is shortsighted, she said.
The U.S. move will also shape what happens elsewhere. The idea behind Covax is to discourage hoarding and focus on vaccinating high-risk people in every country first, a strategy that could lead to better health outcomes and lower costs, experts said.
U.S. nonparticipation makes that harder. “When the U.S. says it is not going to participate in any sort of multilateral effort to secure vaccines, it’s a real blow,” said Suerie Moon, co-director of the Global Health Center at the Graduate Institute of International and Development Studies in Geneva.
“The behavior of countries when it comes to vaccines in this pandemic will have political repercussions beyond public health,” she added. “It’s about, are you a reliable partner, or, at the end of the day, are you going to keep all your toys for yourself?”
Some members of the Trump administration were interested in a more cooperative approach but were ultimately overruled.
Health and Human Services Secretary Alex Azar and Deputy Secretary of State Stephen Biegun had interest in exploring some type of role in Covax, a senior administration official said, speaking on the condition of anonymity because they were not authorized to discuss the decision-making.
But there was resistance in some corners of the government and a belief that the United States has enough coronavirus vaccine candidates in advanced clinical trials that it can go it alone, according to the official and a former senior administration official who learned about it in private discussions.
The question of who wins the race for a safe vaccine will largely influence how the administration’s “America first” approach to the issue plays out.
An unlikely worst-case scenario, experts said, is that none of the U.S. vaccine candidates are viable, leaving the United States with no option since it has shunned the Covax effort.
Another possibility is that a U.S. vaccine does pan out, but the country hoards doses, vaccinating a large number of Americans, including those at low risk, while leaving other countries without.
Experts in health security see at least two problems with this strategy: The first is that a new vaccine is unlikely to offer complete protection to all people, meaning that a portion of the U.S. population will still be vulnerable to imported cases — especially as tourism and trade resume.
The second, related problem is that a U.S. recovery depends on economic recovery elsewhere. If large parts of the world are still in lockdown, the global economy is smarting and supply chains are disrupted, the United States will not be able to bounce back.
“We will continue to suffer the economic consequences — lost U.S. jobs — if the pandemic rages unabated in allies and trading partners,” said Thomas J. Bollyky, a senior fellow at the Council on Foreign Relations and the director of its global health program.
Proponents of a multilateral approach to global public health would like to see all countries coordinate through Covax. Perhaps unsurprisingly, interest is strongest from poor countries, while some larger economies are cutting deals directly with drugmakers.
WHO officials have argued that countries need not choose — they can pursue both strategies by signing bilateral deals and also joining Covax.
“By joining the facility at the same time that you do bilateral deals, you’re actually betting on a larger number of vaccine candidates,” Mariângela Simao, a WHO assistant director for drug and vaccine access, said at an Aug. 17 briefing.
If nothing else, the United States could pledge surplus vaccine doses to Covax to ensure they are distributed in a rational and equitable way, experts said.
Some cautioned against a focus on “winning” the race. Given the complexity of supply chains, vaccine development will necessarily be a global effort, regardless of whether countries want to cooperate.
The decision to steer clear of Covax comes at a time of tremendous change for health diplomacy.
The United States has long been the biggest donor to the WHO and a major funder of vaccine initiatives.
In the early days of the coronavirus pandemic, Trump praised both China and the WHO for their handling of the outbreak. But as the crisis intensified in the United States, he turned on the U.N. health agency.
In April, he announced a freeze on new U.S. funding. Not long after, the State Department started stripping references to the WHO from fact sheets and rerouting funds to other programs.
By July, the administration had sent a letter signaling its intent to withdraw from the WHO.
But untangling the United States from the agency it helped found and shape is not simple — and the terms of the separation are still being assessed.
It is not yet clear, for instance, whether a U.S. withdrawal means the United States will just stop its contributions to the WHO or whether it will stop funding any initiative linked to the agency in any way.
For instance, the White House no longer wants to work with the WHO, but the United States is a major supporter of Gavi, which co-leads the Covax project.
Asked to comment on the Covax decision, a State Department spokeswoman pointed to U.S. funding for Gavi, as well as money for such programs as UNICEF and the Global Fund to Fight AIDS, Tuberculosis and Malaria.
J. Stephen Morrison, director of the Global Health Policy Center at the Center for Strategic and International Studies, said the White House could still reverse course and join Covax, or at least let the Senate fund through Gavi — a political workaround.
“This just shows how awkward, contradictory and self-defeating all of this,” he said. “For the U.S. to terminate its relationship with the WHO in the middle of a pandemic is going to create an endless stream of self-defeating moments.”
U.S. advisory group lays out detailed recommendations on how to prioritize Covid-19 vaccine

A new report that aims to prioritize groups to receive Covid-19 vaccine lays out detailed recommendations on who should be at the front of the line, starting with health care workers in high-risk settings, followed by adults of any age who have medical conditions that put them at significantly higher risk of having severe disease.
Also toward the front of the line would be older adults living in long-term care homes or other crowded settings.
The draft report, which runs 114 pages, was released Tuesday by the National Academies of Sciences, Engineering, and Medicine, which was tasked with the work by Francis Collins, director of the National Institutes of Health, and Robert Redfield, director of the Centers for Disease Control and Prevention.
A virtual public meeting on the recommendations will be held Wednesday afternoon, and the committee’s final report will be submitted later in September.
When Covid-19 vaccines are approved for use, initial supplies will be tight — potentially in the tens of millions of doses. Most of the vaccines under development will require two doses per person: a priming dose followed by a booster either three or four weeks later.
The report suggests that a second phase of vaccinations should involve critical risk workers — people in industries essential to the functioning of society — as well as teachers and school staff; people of all ages with an underlying health problem that increases the risk of severe Covid-19; all older adults not vaccinated in the first phase; people in homeless shelters and group homes, and prisons; and staff working in these facilities.
Young adults, children, and workers in essential industries not vaccinated previously would make up the third priority group. Remaining Americans who were not vaccinated in the first three groups would be offered vaccine during a fourth and final phase.
The report is meant to serve as a guide for more detailed prioritization plans on the order in which Americans will be offered vaccine. That more granular work is already being conducted by the Advisory Committee on Immunization Practices, an expert panel that crafts vaccination guidance for the CDC, and by state, local, and tribal health authorities, who must identify the actual people in their regions who fall into the priority groups.
There has been discussion of prioritizing people of color, who have been disproportionately badly hit in this pandemic. But the report does not recommend that Black, Hispanic, Latinx people, or American Indians or Alaskan natives be treated as a distinct priority group.
The committee suggested that there does not appear to be a biological reason for why these communities are more seriously affected by the pandemic. Instead, it argues, the high rates of infections and deaths in these communities are due to systemic racism that leads to higher levels of poor health and socioeconomic factors such as working in jobs that cannot be done from home or living in crowded settings.
The report therefore prioritized other factors — people with underlying medical problems, people living in crowded environments, for instance — rather than creating priority categories for racial or ethnic groups.
The ACIP’s recommendations will go to the CDC. It remains unclear, however, whether the CDC, Operation Warp Speed — the task force set up to fast-track development of Covid-19 vaccines, drugs and diagnostics — or the White House will make the final determinations on who will be vaccinated first.
The task of determining who should be at the front of the vaccines line is not an easy one, and must be made without key pieces of information. It’s not yet known how many vaccines will prove to be successful, when they will be approved for use and in what quantities. Critically, some vaccines may prove to be more effective in key groups — the elderly, for instance — than others. Knowing that in advance could influence the recommendations, but people working on the priority groups cannot wait for that information to become available.
Initial discussions suggest large numbers of Americans would qualify as members of priority groups, a reality that will likely require additional tough decisions to be made.
CDC estimates that there are between 17 million and 20 million health care workers in the country, and roughly 100 million people with medical conditions that put them at increased risk of severe illness if they contract Covid-19. There are roughly 53 million Americans aged 65 and older and 100 million people in jobs designated as essential services. There is some overlap among these groups — health workers, for instance, are also essential workers.
A report released last month by the Johns Hopkins Center for Health Security recommended dividing priority groups into two tiers, with health workers and others essential to the Covid-19 response in the first tier and other health workers in the second.
In that report, people at greatest risk and their caregivers, and workers most essential to maintaining core societal functions would also be designated to be in the first tier.
Health policy scholars critiqued the Trump campaign’s broad strokes healthcare agenda for his potential second term. While some found it overly vague, even dishonest, one suggested it was precisely what voters want.
Released Sunday night as a list of bullet points, the “Fighting for You” agenda will apparently serve as the Republican platform for the 2020 election. The GOP’s platform committee voted over the weekend to dispense with the customary detailed policy document for this cycle, in favor of simply backing President Trump’s agenda.
That agenda, which the Trump campaign promised would be fleshed out in future speeches and statements, included the following points relevant to healthcare:
Eradicate COVID-19
Healthcare
Reliance on China
Joseph Antos, PhD, a resident scholar in healthcare and retirement policy at the American Enterprise Institute, characterized Trump’s strategy as “Don’t explain it. Just say what your goals are.”
He applauded the brevity of the document, 6 pages in total, covering 10 different policy areas from jobs to healthcare to immigration, as a “smart strategy.”
Voters don’t want to read lengthy policy briefs and gave the “Biden-Sanders Unity Task Force Recommendations” which were over 100 pages long and “unbelievably complicated stuff” as an example of how not to reach voters.
“I think [Trump] got it right. He’s not running a think tank…. He’s running for office. He does have a keen eye for what the average voter could stand to listen to.”
Gail Wilensky, PhD, an economist and senior fellow at Project Hope in Bethesda, Maryland, and CMS administrator under President George H.W. Bush, agreed that a platform packed with policy details doesn’t sway many voters.
This election, she said, is about one thing only: “Trump or not Trump.”
Whither the ACA?
Nevertheless, the Trump campaign’s goals merit attention, often for what they don’t include as well as what they do.
As for the substance of the agenda, the key difference between the Trump administration’s proposed agenda and that of the Democratic nominee, former Vice President Joe Biden, is that the latter aims to expand access to health insurance using the Affordable Care Act’s (ACA) framework, said Wilensky.
While Trump’s 2016 healthcare agenda centered around repealing the ACA, his second-term agenda doesn’t mention the law by name.
Wilensky said she’s glad that Trump did not include ACA repeal among his goals, given that “there’s no historical precedence” for eliminating the core benefits of such far-reaching legislation, now on the books for 10 years and fully implemented for 6.
Kavita Patel, MD, a primary care physician and Brookings Institution scholar in Washington, D.C., who was an advisor on the Democrats’ platform, said, “This is all just posturing and politics and almost a continuation of things [Trump’s] been saying without any real details behind it.”
Many of these items — such as ending surprise billing, lowering health insurance premiums, and cutting prescription drug prices — would have Democrats’ support “but they would get there in a different way,” Patel said.
One thing she was surprised not to see in the agenda were references to abortion or other reproductive health issues, she noted.
Insurance Coverage Neglected
Rosemarie Day, founder and CEO of Day Health Strategies and author of Marching Toward Coverage: How Women can Lead the Fight for Universal Healthcare, was dumbfounded by the overall lack of substance in the agenda, and particularly by the absence of a plan to deal with rising rates of uninsurance related to the pandemic.
Day thought the Trump campaign could have at least included a plan for returning to the “baseline” on the number of uninsured. Another administration might have chosen to promote Medicaid coverage or encourage unemployed workers to enroll on the health insurance exchanges, but not this administration, she said.
“So, they’re really just leaving people out in the cold,” Day said.
Wilensky, too, suggested it would have been “useful” for the Trump campaign to have “talked about how they envision getting more people covered.”
Paul Ginsburg, PhD, director of the USC-Brookings Schaeffer Initiative for Health Policy, said much of the agenda is “just aspirations.”
“‘Put patients and doctors in charge of our healthcare system’? … I don’t know what the policy is, [but] who’s going to quarrel with that?”
Lowering healthcare premiums also sounds “nice” but how that would be achieved is unclear, he said.
One agenda item in the document that really really irked Day was the Trump administration’s pledge to protect people who have pre-existing conditions.
“I consider the ‘covering all pre-existing conditions’ an outright lie,” she said. “I find it incredibly upsetting that [Trump] continues to say that” because he spent his first term attacking the ACA, which does protect pre-existing condition coverage.
Day also noted that the administration has repeatedly promised an ACA replacement without ever delivering an actual proposal.
Responding to the Pandemic
The Trump campaign agenda lists “eradicate COVID-19” on its bullet list, but Patel said it’s “probably not an achievable goal.” A more realistic target is to control it better.
“We have deaths every year and hospitalizations from influenza, but we have a vaccine and we have … strategies to protect people like seniors and young children,” Patel said. “That’s exactly the kind of attitude we have to take” with regard to COVID-19.
For both Patel and Ginsburg, “return to normal” is another aspiration that’s beyond the government’s power to deliver.
“So much depends on a vaccine and its acceptance and how quickly it can be produced,” Ginsburg said.
As for making all critical medicines and supplies for healthcare workers in the United States, Ginsburg acknowledged that it’s theoretically doable, but still unrealistic because it would be “way too expensive.”
“Brand name drugs are routinely produced in other countries as well as the U.S.; I wouldn’t want to upset that supply chain, especially for drugs that are in shortage,” he said.
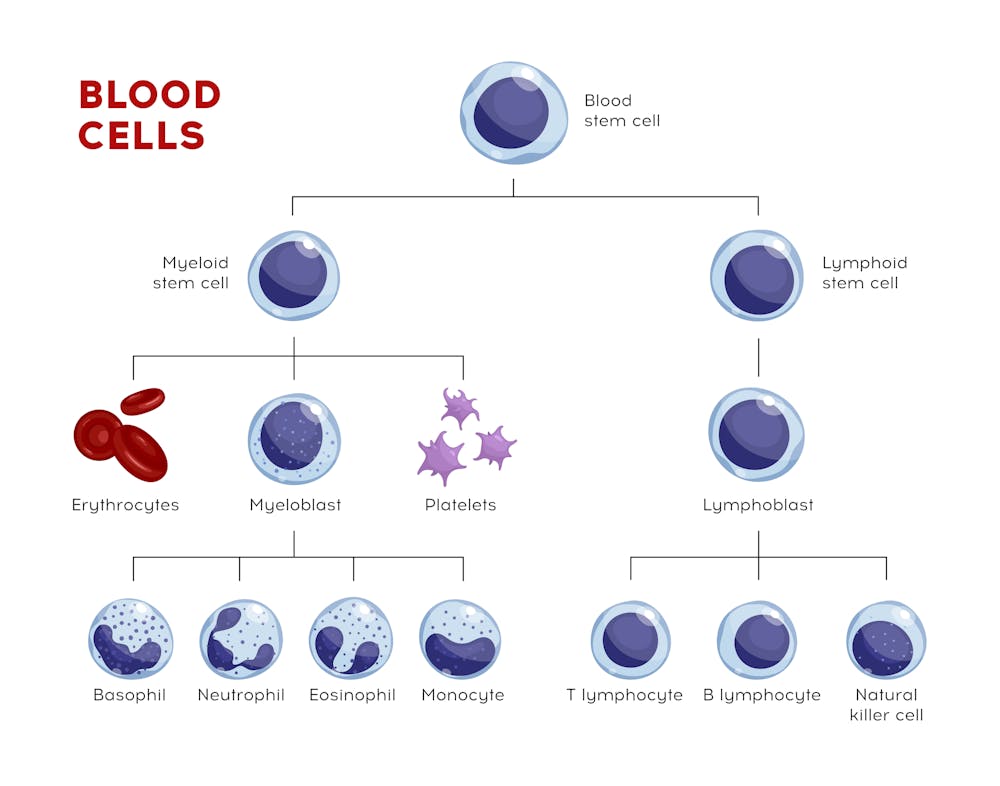
Most people are aware that testing for antibodies in a person’s blood can show if someone has had a specific disease, such as COVID-19. Those antibodies provide protection from getting the disease again.
But in a paper published in the New England Journal of Medicine, researchers found that antibody levels decline in individuals who have recovered from COVID-19, dropping by half every 36 days. Does that mean people who have recovered from COVID-19 have lost their immunity?
I am a geneticist interested in innate immune response – the part of the immune system that we have at birth – and how the innate immune cells “educate” antibody-producing cells about a pathogen and how to identify and destroy it. As I’ll explain, antibodies are important for immunity, but they aren’t the only factor that counts.
The immune system is made up of two parts: innate immunity and adaptive – or acquired – immunity.
The innate immune system, which includes white blood cells called dendritic cells, monocytes and neutrophils, is present at birth and responds instantly to invaders. This group of white blood cells bombard pathogens with destructive chemicals and swallow and destroy viruses and bacteria. The innate immune system provides an instantaneous reaction to a pathogen. The problem is that it’s a blunt instrument – it responds the same way to all perceived threats.
The adaptive immune system, which is made up of B cells and T cells, must learn about a pathogen and its characteristics from the innate immune cells. This system takes longer to kick in, but the up side is that it is very specific and in many cases lasts a lifetime.
The history of pathogen exposure is carried in so-called memory T cells and memory B cells. When an infection is defeated and gone, these cells reside in the peripheral tissues of the body such as lymph nodes or spleen and serve as a memory of the disease-causing virus. This immunological memory is responsible for the host defense and kicks into action in case of the second wave or attack of the pathogen.
It is normal for antibody levels to decline after a person has recovered from a disease. But the New England Journal of Medicine paper raised concerns because it suggests that we are losing our immunological memory – which is as bad as losing a real memory.
B cells and antibodies are only part of the immune response. T cells help B cells produce antibodies – which are proteins that can bind to a specific pathogen and destroy it.
The way this happens is that first the B cells swallow the virus and start producing antibodies.
T cells cannot swallow the virus. But a type of white blood cell called an antigen-presenting cell can. After it does, it “shows” different parts of the virus to the T cells. The T cells then learn about the virus which they can now seek and destroy.
T cells also stick to the B cells and send them the activation signals that help B cells ramp up antibody production.
It suggests that when there are fewer antibodies in the blood, there is a greater chance that a number of individual virus particles, called virions, will survive and escape destruction. Therefore, the remaining virions will continue to proliferate and cause disease.
Herd immunity refers to a population and occurs when a sufficiently high number of people within a community are immune to the virus and incapable of transmitting it. That provides protection for those who are still vulnerable. For example, if 60% of people are protected against COVID – because they have survived the infection and carry antibodies – it might protect (via less frequent interactions) the remaining 40% from getting sick.
But the results in the New England Journal of Medicine suggest that people with lower levels of antibody may still have the virus and may not have symptoms of the disease.
That means that if these people with low antibody levels hang around healthy, uninfected people, they present a danger to them because they can transmit the virus.
In general, the answer is no. If the virus attempts to cause a second infection, the memory B and T cells are able to recognize it, multiply million of times and defend the body against the virus, preventing it from triggering another full-blown infection.
The protection provided by memory T and B cells is the reason that vaccine-based protection works.
However, there are exceptions. A lifelong vaccine against the flu does not work because flu’s genetic code changes rapidly, altering the appearance of the flu, and therefore requires a new vaccine every season.
But with SARS-CoV-2, the problem as I see it, seems to be that those memory T cells and B cells seem to be wiped out.
Antibodies are proteins and last for only between three and four weeks in the blood circulation. To keep antibody levels high, B cells need to replenish them with a fresh supply. But in COVID-19, the declining antibody levels suggest that the cells that produce these antibodies are not present in sufficient numbers, which would explain the drop in antibody levels. Studies of how long immunity from COVID-19 last may shed more light, but for now we do not know the reason why.

With the coronavirus still spreading widely, it’s time to start thinking seriously about influenza, which typically spreads in fall and winter. A major flu outbreak would not only overwhelm hospitals this fall and winter, but also likely overwhelm a person who might contract both at once.
Doctors have no way of knowing yet what the effect of a dual diagnosis might be on a person’s body, but they do know the havoc that the flu alone can do to a person’s body. And, we know the U.S. death toll of COVID-19 as of Aug. 17, 2020 was 170,000, and doctors are learning more each day about the effects of the disease on the body. Public health officials in the U.S. are therefore urging people to get the flu vaccine, which is already being shipped in many areas to be ready for September vaccinations.
Flu cases are expected to start increasing early in October and could last late into May. This makes September and early October the ideal time to get your flu shot.
But there’s reason to be concerned that flu vaccination rates could be lower this year than in past years, even though the risk of getting seriously ill may be higher because of widespread circulation of the coronavirus.
In an effort to avoid getting sick, millions of Americans avoided seeing their health care provider the past few months. Social distancing and stay-at-home orders have resulted in a decreased use of routine medical preventive services such as vaccinations. Many employers that often provide the flu shot at no cost to employees are allowing employees to work from home, potentially limiting the number of people who will get the flu shot at their jobs.
As a health care professional, I urge everyone to get the flu vaccine in September. Please do not wait for flu cases to start to peak. The flu vaccine takes up to two weeks to reach peak effectiveness, so getting the vaccine in September will help provide the best protection as the flu increases in October and later in the season.
Both COVID-19 and the flu are contagious respiratory illness that present with similar symptoms. Both viruses can impact the elderly and those with certain chronic conditions, such as heart and lung disease, the hardest.
Data on flu vaccination rates from 2018-2019 show that only 49% of Americans six months of age and older received the flu vaccine. The vaccine’s effectiveness varies each season, with early data from the 2019-2020 flu season indicating a vaccine effectiveness rate of 50% overall, and 55% in youth.
While some may think this effectiveness rate is low, the flu vaccine remains the single best way to prevent the flu and related complications. For example, during the 2018-2019 flu season, flu vaccination was estimated to prevent 4.4 million flu illnesses, 58,000 flu hospitalizations and 3,500 deaths. Early data from the 2019-2020 flu season estimates there were 39-56 million flu illnesses, 18-26 million flu-related medical visits, 410-740,000 hospitalizations and up to 62,000 deaths. Much of this disease burden is preventable from higher flu vaccination rates.
It is now quite apparent that COVID-19 will still be circulating during flu season, which makes getting a flu vaccine more important than ever. As schools, our communities and our economy continue to reopen, it is vital to get the flu vaccine for personal, family and community protection.

A flu camp in Lawrence, Maine during the 1918 influenza pandemic. Nurses and doctors tried desperate measures to stop the spread of the disease, which ultimately killed more than 675,000 people in the U.S. alone. Bettmann/Getty Images
Severe cases of both COVID-19 and the flu require the same lifesaving medical equipment. This highlights the importance of getting the flu vaccine for not only your own personal health but also the health of your community. Receiving the flu vaccine will help reduce the burden of respiratory illness on our already very overstretched health care system. By increasing flu vaccination rates, we can reduce the overall impact of respiratory illnesses on the population and hence lower the resulting burden on the health care system during the COVID-19 pandemic.
Because flu vaccination protects against one of these respiratory illnesses, the CDC recommends everyone (with few exceptions) six months of age and older get an annual flu vaccine. While the flu vaccine will not protect you against COVID-19, the flu vaccine will reduce your risk of developing the flu as well as reduce your risks of flu-related complications including hospitalization and even death.
While it may seem like there is so much out of our control during this pandemic, getting the flu vaccine, practicing proper hand washing, social distancing and wearing face coverings are within our control and will protect not only you but also your family and community.
If you are not getting the flu vaccine from your employer, think about alternative sources now. Vaccines should be available in most areas by Sept. 1.
Mainly, make sure you take advantage of this potentially lifesaving vaccine. Get it on your calendar for early September now. And remember, the flu shot cannot give you the flu.

There’s no vaccine for Covid-19, but there’s one for influenza. With the season’s first doses now shipping, officials are struggling over how to get people to take it.
As public health officials look to fall and winter, the specter of a new surge of Covid-19 gives them chills. But there is a scenario they dread even more: a severe flu season, resulting in a “twindemic.”
Even a mild flu season could stagger hospitals already coping with Covid-19 cases. And though officials don’t know yet what degree of severity to anticipate this year, they are worried large numbers of people could forgo flu shots, increasing the risk of widespread outbreaks.
The concern about a twindemic is so great that officials around the world are pushing the flu shot even before it becomes available in clinics and doctors’ offices. Dr. Robert Redfield, director of the U.S. Centers for Disease Control and Prevention has been talking it up, urging corporate leaders to figure out ways to inoculate employees. The C.D.C. usually purchases 500,000 doses for uninsured adults but this year ordered an additional 9.3 million doses.
Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, has been imploring people to get the flu shot, “so that you could at least blunt the effect of one of those two potential respiratory infections.”
In Britain, Prime Minister Boris Johnson has been waging his own pro flu-shot campaign. Last month, he labeled people who oppose flu vaccines “nuts” and announced the country’s largest ever rollout of the shots. In April, one of the few reasons Australia allowed citizens to break the country’s strict lockdown was to venture out for their flu shots.
The flu vaccine is rarely mandated in the U.S. except by some health care facilities and nursery schools, but this month the statewide University of California system announced that because of the pandemic, it is requiring all 230,000 employees and 280,000 students to get the flu vaccine by November 1.
A life-threatening respiratory illness that crowds emergency rooms and intensive care units, flu shares symptoms with Covid-19: fever, headache, cough, sore throat, muscle aches and fatigue. Flu can leave patients vulnerable to a harsher attack of Covid-19, doctors believe, and that coming down with both viruses at once could be disastrous.
The 2019-20 flu season in the United States was mild, according to the C.D.C. But a mild flu season still takes a toll. In preliminary estimates, the C.D.C. says that cases ranged from 39 million to 56 million, resulting in up to 740,000 hospitalizations and from 24,000 to 62,000 flu-related deaths.
According to the C.D.C., flu season occurs in the fall and winter, peaking from December to February, and so was nearing its end as the pandemic began to flare in the United States in March.

But now, fighting flu proactively during the continuing pandemic presents significant challenges: not only how to administer the shot safely and readily, but also how to prompt people to get a shot that a majority of Americans have typically distrusted, dismissed and skipped.
With many places where the flu shot is administered en masse now inaccessible — including offices and plants that offered it free to employees on site and school health clinics — officials have been reaching out to local health departments, health care providers and corporations to arrange distribution. From now through Oct. 31, publicity campaigns will blast through social media, billboards, television and radio. Because the shot will be more difficult to access this year, people are being told to get it as soon as possible, although immunity does wane. There will be flu shot tents with heaters in parking lots and pop-up clinics in empty school buildings.
Because of the efforts, vaccine makers are projecting that a record 98 million flu shots will be given this year in the United States, about 15 percent more than doses ordered last year. The Kaiser Permanente health care system will be flooding more than 12 million of its members with flu shot reminders via postcard, email, text and phone calls.
Pharmacies and even supermarkets are expected to play a bigger role than they have in previous years. As of this week, Walgreens and CVS will have flu shots available. Walgreens will be hosting additional off-site flu vaccine clinics in community centers and churches. To reduce contact time, CVS is allowing patients to fill out paperwork digitally.
In New York City, which averages about 2,000 flu-related deaths a year, the health department has been reaching out to hundreds of independent pharmacies to administer the shots, because they are often located in outer-borough neighborhoods where the coronavirus has been rampaging. The health department has a detailed online flu vaccine locator.
“Access is a problem for all adult vaccines,” said L. J. Tan, chief strategy officer for the Immunization Action Coalition, a nonprofit group that works to increase vaccination rates, who was an early promoter of the term twindemic. “Adults may think, If I can get the flu shot easily, I might consider it.”
But as difficult as getting the flu shot to people safely will be, perhaps harder still will be persuading them to actually get it. In the 2018-19 flu season in the United States, only 45.3 percent of adults over 18 got the vaccine, with rates for those ages 18 to 50 considerably lower.
Skepticism to this vaccine runs high, particularly in communities of color because of longstanding distrust and discrimination in public health. A 2017 study in the journal Vaccine noted that, compared with white people, “African Americans were more likely to report barriers to vaccination, were more hesitant about vaccines in general and the flu vaccine specifically, more likely to believe in conspiracy theories and use naturalism as an alternative to getting vaccinated.”
Across all demographic groups, perhaps the most striking reason given for avoiding the flu vaccine is that people do not see it as efficacious as, say, the measles vaccine.
Indeed, it is a good vaccine but not a great one. It must be repeated annually. Immunity takes up to two weeks to kick in. But its efficacy also depends on how accurately infectious disease centers worldwide forecast which strains are expected to circulate in the coming year. And then those strains can mutate.
Although the flu shot confers immunity at all ages over six months, it can be less complete in people over 65. Depending on many factors, the shot’s effectiveness in a given year can range from 40 to 60 percent.
“But a vaccine not given won’t protect anyone,” said Dr. Jane R. Zucker, assistant commissioner for the Bureau of Immunization at the New York City Health Department, which has been hosting webinars for providers about how to have conversations about the flu shot with hesitant patients.
As health officials note, should a vaccinated person contract the flu, the severity will almost certainly be reduced, hospitalization rarely necessary. Especially with Covid-19 raging, public officials reason, those odds look pretty good.
Another reason people give for not getting the shot is they think it makes them sick.
“People who say ‘I’ll never get it because it gives me the flu’ have not had the flu and don’t know what it is,” said Patsy Stinchfield, senior director of infection prevention at Children’s Minnesota.
“What you’re feeling is your body’s immune response to the virus’s antigens,” said Ms. Stinchfield, a member of the C.D.C.’s influenza work group. “You may feel flu-ish. And that’s a good thing. It’s your body’s way of saying, ‘I am ready for the flu, and I won’t get as sick if I get the real one.’”
Public campaigns will describe the shot as a critical weapon during the pandemic. “Hopefully people will say, ‘There’s no Covid vaccine so I can’t control that, but I do have access to the flu vaccine and I can get that,’” Ms. Stinchfield said. “It gives you a little power to protect yourself.”
Other campaigns will emphasize familial and community responsibility.
Usually, flu vaccine compliance rates among people ages 18 to 49 are low. Vermont’s, for example, is only about 27 percent.
Christine Finley, the state’s immunization program manager, believes that rates will improve because of the pandemic’s stay-at-home households. “People are more aware that the risks they take can negatively impact others,” she said. “They’re often taking care of young children and older parents.”

If any example could prove instructive about protective behavior and flu vaccines during the coronavirus epidemic, it could well be Australia.
Australia’s flu vaccine rate tends to be modest, but this year demand was high. The government’s rollout of the shot began earlier than usual for the June-through-August winter because the coronavirus pandemic was exploding. Though the government had also issued strict no-entry limits among many states and territories and bans on international travel, the flu shot was one of the few reasons people could emerge from lockdown.
The prevalent strain circulating in the country is Type A, the most common and virulent form of flu, said Dr. Kelly L. Moore, a public health expert at the Vanderbilt University School of Medicine.
According to the C.D.C., Type A is the most likely to circulate globally. It mutates readily, particularly as it jumps between animals and humans.
“There are two strains of Type A influenza in the vaccine,” Dr. Moore said, “and so the very best way to protect yourself is to get the shot.”
Reported cases of flu in Australia have dropped 99 percent compared with 2019.
“Australia’s milder-than-usual flu season is likely the result of a number of factors — strong flu vaccination uptake, social distancing, but also severely decreased movement of people,” said Dr. Jonathan Anderson, a spokesman for Seqirus, a supplier of flu vaccine.
But though American public health authorities usually look to Australia’s flu season as a predictive, Australians say this year it’s not a reliable indicator.
“This situation is of no comfort as these measures do not apply to the United States where the populace has never been effectively physical distancing,” nor have the country’s entry restrictions been as onerous, said Dr. Paul Van Buynder, a public health professor at Griffith University in Queensland, Australia.
All that Americans can do is get vaccinated against flu, he added, because circulation of the coronavirus remains high.
“It is likely they will have a significant influenza season this northern winter,” he said.