
Merry Christmas



A flurry of European travel restrictions announced Sunday over worries about a fast-spreading new variant of the coronavirus have spurred curiosity and concern that the mutation could infect Americans.
After officials in the United Kingdom said Saturday that the variant first identified there was spreading 70 percent faster than others, Google searches about the mutation have spiked. But American public health experts and federal officials say that although it appears that the variant may be more contagious, it is not any more dangerous than others already detected in the United States.
“I don’t think there should be any reason for alarm right now,” Adm. Brett Giroir, who has been in charge of testing, told George Stephanopoulos on ABC’s “This Week.”
Variations to the virus are nothing new, and experts say the novel coronavirus does not mutate as much as influenza, meaning it is less likely that a vaccine would need to be developed every year to keep up with the new strains.
Still, much about this variant remains unknown, such as whether it is a new strain — a functionally different version of the virus.
“The take-home message for right now is that we need to get more information,” said Krutika Kuppalli, an infectious-diseases specialist at the Medical University of South Carolina. “In the meantime, we all need to really double down on our public health measures — wearing masks, remaining physically distanced, avoiding crowds of people.”
In September, U.K. researchers discovered the variant’s prevalence when they collected samples from infected people in southeastern England. It seemed to spread quickly.
“This lineage came up quite rapidly,” Nick Loman, one of the researchers and a professor of microbial genomics at the University of Birmingham, told The Washington Post.

Since then, Australia, Denmark and the Netherlands have identified cases of the variant in their countries, the World Health Organization told the BBC. On Sunday, Italian officials announced that a patient returning from Britain “in the last few days” was in isolation after scientists detected the mutation.
In South Africa, health officials said Sunday that a version of the virus similar to the U.K. variant has been found in 80 to 90 percent of samples analyzed since mid-November.
The virus has not been detected in the United States, but officials are watching for developments in Britain, Giroir told Stephanopoulos.
Although guidance from federal agencies discourages traveling to Britain, the United States has not banned travel from there.
“I really don’t believe we need to do that yet,” Giroir said.
But New York Gov. Andrew M. Cuomo (D) pointed out Sunday that it would take only one flight to the United States to spread the mutation, urging federal officials to restrict travel.
“Right now, this variant in the U.K. is getting on a plane and flying to JFK,” Cuomo said on a conference call with reporters.
However, Kuppalli warned that little is known about the effectiveness of a ban, referring to instances in which people rushed to airports and congregated in long lines trying to travel before restrictions were put into effect earlier in the pandemic.
It is also not entirely certain whether the virus gained its foothold in the southeast of England because of its altered genetic markers or through super-spreading events, said Scott Gottlieb, a former head of the Food and Drug Administration.
“It seems like this new strain is more contagious,” Gottlieb said on CBS’s “Face the Nation.” “It doesn’t seem to be any more virulent, any more dangerous than run-of-the-mill covid. The next question is: Will it obviate our natural immunity?”
As of now, experts say there has been no indication that the variant is resistant to the Pfizer and Moderna vaccines authorized by the FDA.
“There is no reason to believe that the vaccines that have been developed will not be effective against this virus as well,” Vivek H. Murthy, President-elect Joe Biden’s nominee for surgeon general, told host Chuck Todd on NBC’s “Meet the Press.” “The bottom line is if you’re at home and hearing this news, it does not change what we do in terms of precautions.”
Although the vaccine remains out of reach for most Americans, following guidance such as social distancing and wearing masks remains the most practical way to prevent transmission.


https://mailchi.mp/e38b070b8787/the-weekly-gist-december-18-2020?e=d1e747d2d8

It turns out it’s not just the kids who aren’t getting snow days this year. This week, we spoke with an executive at a health system hit hard by Wednesday’s Nor’easter, and asked how the system was faring with the expected 18 inches of snowfall. He replied that the medical group was as busy as usual.
With all the work this spring to expand telemedicine capabilities, clinic staff were able to reach out to patients the day before the storm, and proactively convert a majority of scheduled in-person clinic visits to telemedicine. “Normally we would’ve been closed, and most appointments rescheduled for weeks down the road,” he told us. Instead, they were able to keep most of those visits in their scheduled time slot.
“Now that we have a systemwide process for telemedicine, I don’t think we’ll have a reason for the clinic to take a snow day again.” It’s a clear win-win for the system and patients: patient care seamlessly goes on. It’s easy to see the many use cases for the ability to toggle between in-person and virtual visits. A parent is stuck at home with a sick kid, and can’t make her endocrinologist appointment? Moved to virtual! A patient has an unexpected business trip taking him out of town? Don’t cancel, let’s do that follow-up visit via telemedicine.
We’ve been worried about the slowdown in progress made on telemedicine as patients switched back to in-person visits across the summer and fall. The ability to continue patient care during a record-breaking snowstorm is a perfect illustration of why it’s critical not to “backslide” with virtual care: meeting patients where they are, regardless of circumstances, is an essential part of building long-term loyalty and care continuity.
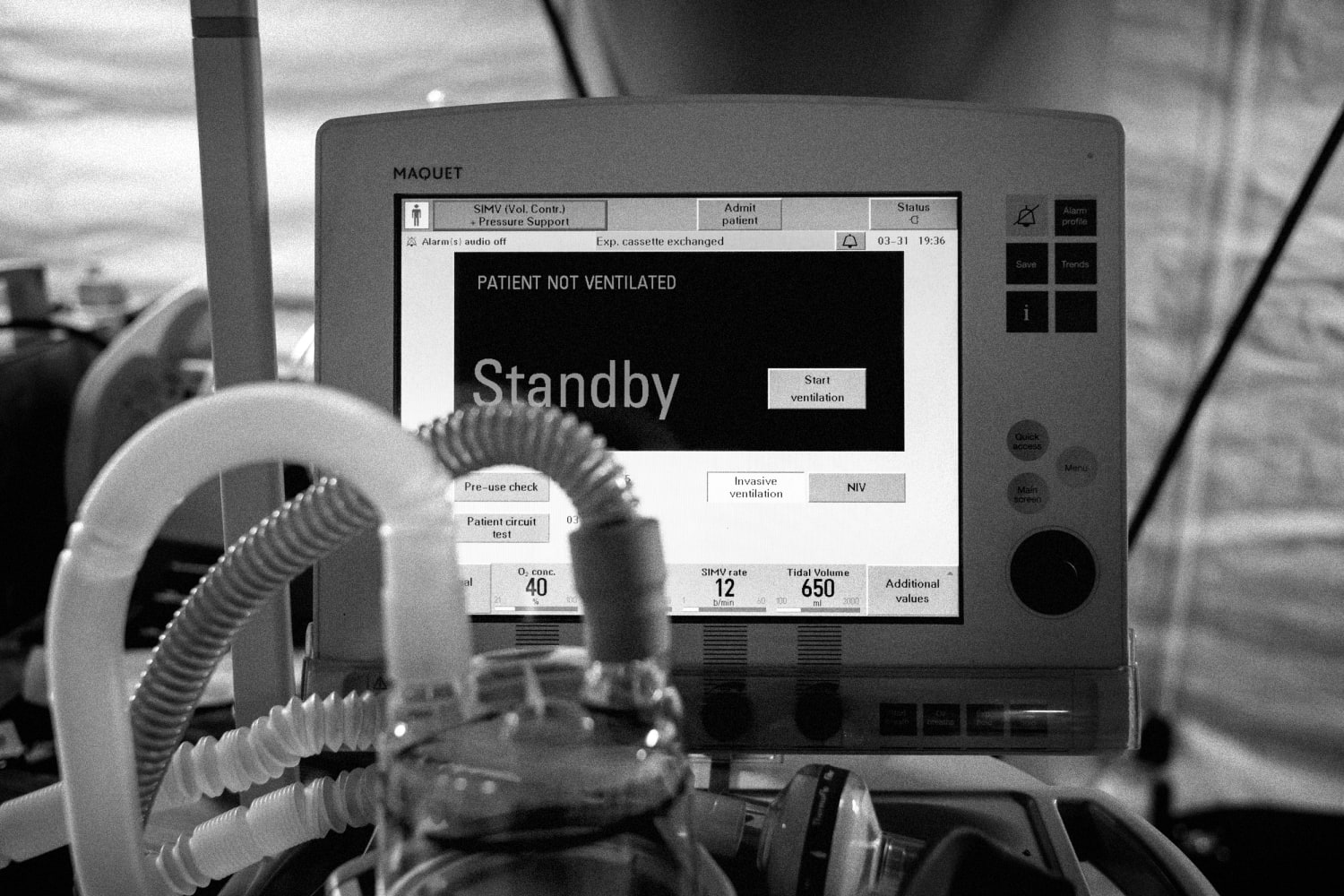
Several hospitals in Los Angeles County are preparing for the possibility of restrategizing care delivery in the coming weeks amid growing COVID-19 hospitalizations, according to the Los Angeles Times.
Los Angeles County reported 6,018 COVID-19 hospitalizations Dec. 20 — a 2.5 percent increase from the day prior — with 1,198 patients in ICUs, according to the state’s data dashboard. Statewide, 17,750 patients are hospitalized with COVID-19, 3,710 of them in ICUs.
The LA Times obtained a document recently circulated among physicians at the four county-run hospitals that outlines resource allocation in crisis situations. The guidelines call for physicians to save as many patients as possible versus trying everything to save a patient, meaning those less likely to survive will not receive the level of care they would have otherwise. L.A. County Health Services Director Christina Ghaly, MD, told the LA Times that the guidelines were not in place as of Friday night.
California activated its “mass fatality” program last week, which coordinates mutual aid across several government agencies when more deaths take place in a period of time than can be handled by local coroner or medical emergency personnel, NPR reports.
Nearly all of the state is under stay-at-home orders, with residents prohibited from gathering with anyone outside their immediate household.
“I have yet to see any clear signals that things are slowing down, and I’m very concerned about the next two months,” Timothy Brewer, MD, an epidemiologist with UCLA Health, told the LA Times. He said UCLA Health is scheduling several infectious disease specialists to be on call at any time, and the biggest issue is that hospitals may quickly run out of providers who can administer ICU-level care.

Congressional leaders have reached an agreement on a $900 billion COVID-19 relief package and $1.4 trillion government funding deal with several healthcare provisions, according to Senate Majority Leader Mitch McConnell, R-Ky., and Minority Leader Chuck Schumer, D-N.Y.
Here are seven things to know about the relief aid and funding deal:
1. Congressional leaders have yet to release text of the COVID-19 legislation, but have shared a few key details on the measure, according to CNBC. Becker’s breaks down the information that has been released thus far.
2. The COVID-19 package includes $20 billion for the purchase of vaccines, about $9 billion for vaccine distribution and about $22 billion to help states with testing, tracing and other COVID-19 mitigation programs, according to Politico.
3. Lawmakers are also expected to include a provision changing how providers can use their relief grants. In particular, the bill is expected to allow hospitals to calculate lost revenue by comparing budgeted revenue for 2020. Hospitals have said this tweak will allow them to keep more funding.
4. The agreement also allocates $284 billion for a new round of Paycheck Protection Program loans.
5. The COVID-19 relief bill also provides $600 stimulus checks to Americans earning up to $75,000 per year and $600 for their children, according to NBC. It also provides a supplemental $300 per week in unemployment benefits.
6. The year-end spending bill includes a measure to ban surprise billing. Under the measure, hospitals and physicians would be banned from charging patients out-of-network costs their insurers would not cover. Instead, patients would only be required to pay their in-network cost-sharing amount when they see an out-of-network provider, according to The Hill. The agreement gives insurers 30 days to negotiate a payment on the outstanding bill. After that period, they can enter into arbitration to gain higher reimbursement.
7. Lawmakers plan to pass the relief bill and federal spending bill Dec. 21.
https://mailchi.mp/e38b070b8787/the-weekly-gist-december-18-2020?e=d1e747d2d8

As the first Americans receive COVID vaccines, supplies remain limited even for the highest-risk populations. And with doses now in the pipeline, states are facing more intense questions about how they will prioritize vaccine delivery across demographic and at-risk groups. The graphic above shows an estimated vaccination timeline, based on the Centers for Disease Control and Prevention’s (CDC) recommended schedule. It illustrates the relative size of different populations in each allocation phase, along with the likely difficulty of targeting them and verifying eligibility. The first phase is divided into three waves (1a, 1b, 1c) for at-risk populations and essential workers, while the second phase includes the rest of the adult population, as well as children (though pediatric clinical trials are still in early stages).
Unsurprisingly, the CDC recommends that those most at risk for infection and severe disease—healthcare workers and nursing home residents—receive the 20M doses available by year’s end. While most states are generally adhering to the initial recommendations on priority groups for phase 1a set by the CDC’s Advisory Committee on Immunization Practices (ACIP), several have made adjustments. At least three are including law enforcement personnel in phase 1a, and others are further categorizing healthcare workers into high-, medium-, and low-risk groups. This weekend, ACIP will reconvene to create its official recommendations for phases 1b and 1c, which include the much larger populations of adults over age 65, and those with high-risk medical conditions.
Beyond eligibility guidelines, larger questions loom. How would someone “verify” that they have a high-risk condition? Who will reach out to older Americans to let them know they are eligible, and where to access the vaccine? As vaccine rollout continues, providers should anticipate the role they will likely play in managing patients “in the queue”, documenting eligible conditions and establishing regular information channels to keep people informed about the current status of vaccine planning and access.
https://mailchi.mp/e38b070b8787/the-weekly-gist-december-18-2020?e=d1e747d2d8

But first, we have a difficult period to get through. This week again saw record-breaking numbers of cases, hospitalizations, and deaths from COVID-19, with Thursday alone bringing more than 238,000 new cases—and a staggering 3,293 fatalities.
Nearly 115,000 Americans are currently hospitalized with COVID, a rise of 16 percent from just two weeks ago, and in many places a precarious capacity situation has turned perilous. Conditions have worsened precipitously in California, with only Tennessee, Oklahoma, and Rhode Island registering more daily COVID cases per 100,000 population than the Golden State, although cases are still on the rise across 80 percent of states and territories.
Intensive care availability in Southern California hit zero, with ICU volume there expected to double or triple by this time next month. The same stresses are playing out in dozens of markets across the country, leading to a staffing sustainability crisis that can’t be solved through paying overtime, cancelling vacations or looking to travel nurses to fill the gaps in a now nationwide crisis. With the Christmas and New Year’s holidays still ahead, experts predict COVID cases won’t peak until sometime in mid-January, with a peak in hospitalizations and deaths following several weeks after.
Several states and cities tightened restrictions on gatherings and issued new stay-at-home orders, in an effort to keep new cases at a level that allows hospitals to manage through the next several weeks and maintain care quality and access for COVID and non-COVID patients alike. The coming weeks will require every American to take greater precautions than at any time during the course of this pandemic.