
Cartoon – History Repeating




R1 RCM plans to pay $30 million for health IT giant Cerner’s revenue cycle business.
Chicago-based R1, a leading revenue cycle management technology vendor, is acquiring Cerner RevWorks’ services business and commercial, nonfederal client relationships. The deal, which was announced Wednesday, does not include RevWorks’ federal clients.
R1 said it plans to hire Cerner RevWorks employees once the deal closes in the third quarter of 2020.
Both companies have committed to a seamless integration between the company’s technology-enabled services platform and Cerner’s software, R1 said in a press release.
As part of the transaction, Cerner said it will extend R1’s revenue cycle capabilities and expertise to Cerner clients and new prospects, helping drive sustainable financial improvements for providers while enhancing their patients’ overall experience.
The closing of the acquisition is expected to take place in the third quarter of 2020, subject to customary closing conditions.
According to R1’s filing with the U.S. Securities and Exchange Commission (SEC), the deal is valued at $30 million inclusive of working capital, financed with cash on the balance sheet.
The acquisition price will be paid in three installments, according to the SEC filing.
R1’s stock rose 12% Wednesday following the news.
The deal further establishes R1’s footprint across the acute and ambulatory markets, the company said in the SEC filing. The RevWorks business brings in approximately $80 million in annual revenue across more than 150 customers.
“We look forward to working collaboratively with Cerner to deliver superior results for healthcare providers and the communities they serve,” said Gary Long, executive vice president and chief commercial officer of R1. “With our interoperable technology and end-to-end platform, we are well-positioned to serve Cerner’s customers, as well as other healthcare organizations across the country.”
“Cerner’s overall goal is to deliver client success and accelerate our ability to deliver scalable innovations,” said Brenna Quinn, senior vice president of revenue cycle management at Cerner, in a statement.
In a statement provided by the company, Cerner executives said the deal with R1 will bring its commercial, nonfederal clients a total solution that pairs “Cerner’s advanced technology with R1’s world-class revenue services, ultimately optimizing financial performance for health systems.”
“Cerner remains committed to and heavily invested in its revenue cycle solutions to help our clients combine clinical, financial, and operational health information when and where it’s needed,” the company said.
Centerview Partners LLC acted as financial adviser, and Kirkland & Ellis LLP acted as legal adviser to R1. Greenhill & Co. acted as an adviser to Cerner.
Earlier this year, R1 acquired SCI Solutions, a provider of SaaS-based scheduling and patient access solutions, for approximately $190 million in cash.
The RCM vendor’s revenue grew 16.2% in the first quarter of 2020, up $44.6 million to reach $320.5 million.
However, during its first-quarter earnings call in May, R1 executives said the company is expecting to see revenue decline by $10 million to $20 million in the second quarter, driven by lower patient volumes for its smaller physician customers.
Cerner’s revenue cycle business took a hit last year when Adventist Health terminated its revenue cycle outsourcing contract with the company, resulting in a $60 million impairment charge for Cerner in the third quarter of 2019, the company reported during its third-quarter earnings call.
Adventist transitioned all its revenue cycle operations to Huron Consulting Group. At the time, about 1,700 Cerner employees transitioned over to Adventist and Huron.
During the company’s fourth-quarter and year-end 2019 earnings call in February, Cerner Chief Financial Officer Marc Naughton hinted at a potential sell-off of the RevWorks business.
“Those areas that we don’t think are the growth areas for the company we want to focus on, we’re going to consider divesting as one of the options,” Naughton said during a Q&A with analysts. “It will be an existing business that we will basically go out to market and look for opportunities to say here is this asset, it’s something we’re willing to let go and some of these assets have significant value.”

OptumRx researchers are highlighting three more drug products that payers should be keeping an eye on in 2020.
Experts said in the pharmacy benefit manager’s second-quarter drug pipeline report echoed expectations from the first quarter that orphan drugs will be a major trend to watch as the year continues. Sumit Dutta, M.D., chief medical officer at OptumRx, wrote in the report these drugs will likely account for close to 40% of Food and Drug Administration approvals this year.
Dutta said Optum is seeing more drug manufacturers jump into developing orphan drug products, which are generally considered less appealing as their market—and thus financial value—is more limited.
“What is new is that we now are starting to see the development of orphan drugs become more competitive, increasing the potential for reduced costs and broader patient accessibility,” he wrote.
In 2018, the FDA approved more orphan drugs than non-orphan drugs for the first time. Optum’s first-quarter report also noted that these products are often pricey, as they target specific conditions. On average, orphan drugs cost $147,000 or more per year.
Of the three products highlighted in the second-quarter report, two are orphan drugs. Here’s more on what Optum’s analysts think payers need to know:
If the FDA gives risdiplam a thumbs-up, it would become the first oral therapy for spinal muscular atrophy (SMA), a rare group of severe neuromuscular disorders. SMA is one of the most common genetic causes for infant mortality and affects about 1 in 11,000 babies.
There are only two treatments for SMA that are currently approved by the FDA, meaning there’s a significant unmet need for therapies, particularly oral medications, Optum said. The other treatments available are Spinraza, which requires repeated spinal injections, and gene therapy Zolgensma, the world’s most expensive drug.
Risdiplam would be administered orally once a day, which would likely draw significant interest from patients and their families, according to the report.
“Practically speaking, the competitive advantage for risdiplam will rest mainly in its oral route of administration, and perhaps, a lower cost,” according to the report.
The analysts did caution that risdiplam is still in clinical trials and while results are promising, long-term outcomes associated with the drug are unclear.
Viltolarsen is in development to treat Duchenne muscular dystrophy (DMD), a rare genetic disease that impacts young boys. There is a large unmet need for drugs to treat DMD, Optum said in the report, as it’s linked with significant sickness and death.
About 6,000 people in the U.S. have this disease, according to the report.
Vitolarsen is an “exon-skipping” drug that “short circuits” the genetic mutations that cause DMD. If approved, it would be the third such drug for the disease, and the second targeting a specific mutation that affects about 8% of those with DMD.
The drug has only been tested in small sample sizes, and there are limited safety data available, according to the report.
Trodelvy, the brand name of an antibody-drug conjugate (ADC) therapy aimed at metastatic triple-negative breast cancer, was approved in April.
Triple-negative breast cancers test negative for the three most common causes of cancer and are thus untreatable by many front-line therapies, though they are treatable by chemotherapy, according to the report. ADC products like Trodelvy combine genetically engineered antibodies and traditional chemotherapy drugs into one intravenous therapy.
Optum is highlighting Trodelvy as it expects ADC medications to be a trend to monitor in the near future, because they could be applied to conditions outside of oncology, according to the report.
“We can think of ADCs as a refinement or extension of precision medicine, which aims at maximizing therapeutic benefits while minimizing undesired side effects for an individual patient,” the researchers wrote. “As the field advances, we can look for new conjugate ‘payloads’ that will go far beyond hunting cancer cells.”
“Various manufacturers are exploring how to leverage the ADC approach to produce vaccines, radiological treatments, immunosuppressive, cardiovascular and more,” they wrote.
Sixty of the largest hospital chains in the U.S., including publicly traded and nonprofit systems, have received more than $15 billion in emergency funds through the Coronavirus Aid, Relief and Economic Security Act, according to an analysis by The New York Times.
Congress has allocated $175 billion in relief aid to hospitals and other healthcare providers to cover expenses or lost revenues tied to the COVID-19 pandemic. The first $50 billion in funding from the CARES Act was distributed in April. Of that pool, HHS allocated $30 billion based on Medicare fee-for-service revenue and another $20 billion based on hospitals’ share of net patient revenue. HHS also sent $12 billion to hospitals that provided inpatient care to large numbers of COVID-19 patients and $10 billion to hospitals and other providers in rural areas.
Though one of the goals of the CARES Act was to avoid job losses, at least 36 of the largest hospital systems that received emergency aid have furloughed, laid off or reduced pay for workers, according to the report.
Approximately $1.7 billion in bailout funds went to eight large nonprofit health systems: Mayo Clinic in Rochester, Minn.; Trinity Health in Livonia, Mich.; Beaumont Health in Southfield, Mich.; Henry Ford Health System in Detroit; SSM Health in St. Louis; Mercy in St. Louis; Fairview Health in Minneapolis; and Prisma Health in Greenville, S.C. Mayo Clinic furloughed or cut hours of about 23,000 workers, and the other seven health systems furloughed or laid off a total of more than 30,000 employees in recent months, according to The New York Times.
The pandemic has taken a financial toll on hospitals across the U.S. They’re losing more than $50 billion per month, according to a report from the American Hospital Association. Of the eight nonprofit systems that collected $1.7 billion in relief aid, several have reported losses for the first quarter of this year, which ended March 31. For instance, Mayo Clinic posted a $623 million net loss, SSM Health’s loss totaled $471 million, and Beaumont and Henry Ford Health System reported losses of $278 million and $235 million, respectively.
Since CARES Act payments were automatically sent to hospitals, some health systems have decided to return the funds. Kaiser Permanente, a nonprofit system, is returning more than $500 million it received through the CARES Act. The Oakland, Calif.-based health system ended the first quarter with a $1.1 billion net loss.
Access the full article from The New York Times here.

Seriously ill people avoided hospitals and doctors’ offices. Patients need to return. It’s safe now.
More than 100,000 Americans have died from Covid-19. Beyond those deaths are other casualties of the pandemic — Americans seriously ill with other ailments who avoided care because they feared contracting the coronavirus at hospitals and clinics.
The toll from their deaths may be close to the toll from Covid-19. The trends are clear and concerning. Government orders to shelter in place and health care leaders’ decisions to defer nonessential care successfully prevented the spread of the virus. But these policies — complicated by the loss of employer-provided health insurance as people lost their jobs — have had the unintended effect of delaying care for some of our sickest patients.
To prevent further harm, people with serious, complex and acute illnesses must now return to the doctor for care.
Across the country, we have seen sizable decreases in new cancer diagnoses (45 percent) and reports of heart attacks (38 percent) and strokes (30 percent). Visits to hospital emergency departments are down by as much as 40 percent, but measures of how sick emergency department patients are have risen by 20 percent, according to a Mayo Clinic study, suggesting how harmful the delay can be. Meanwhile, non-Covid-19 out-of-hospital deaths have increased, while in-hospital mortality has declined.
These statistics demonstrate that people with cancer are missing necessary screenings, and those with heart attack or stroke symptoms are staying home during the precious window of time when the damage is reversible. In fact, a recent poll by the American College of Emergency Physicians and Morning Consult found that 80 percent of Americans say they are concerned about contracting the coronavirus from visiting the emergency room.
Unfortunately, we’ve witnessed grievous outcomes as a result of these delays. Recently, a middle-aged patient with abdominal pain waited five days to come to a Mayo Clinic emergency department for help, before dying of a bowel obstruction. Similarly, a young woman delayed care for weeks out of a fear of Covid-19 before she was transferred to a Cleveland Clinic intensive care unit with undiagnosed leukemia. She died within weeks of her symptoms appearing. Both deaths were preventable.
The true cost of this epidemic will not be measured in dollars; it will be measured in human lives and human suffering. In the case of cancer alone, our calculations show we can expect a quarter of a million additional preventable deaths annually if normal care does not resume. Outcomes will be similar for those who forgo treatment for heart attacks and strokes.
Over the past 12 weeks, hospitals deferred nonessential care to prevent viral spread, conserve much-needed personal protective equipment and create capacity for an expected surge of Covid-19 patients. During that time, we also have adopted methods to care for all patients safely, including standard daily screenings for the staff and masking protocols for patients and the staff in the hospital and clinic. At this point, we are gradually returning to normal activities while also mitigating risk for both patients and staff members.
The Covid-19 crisis has changed the practice of medicine in fundamental ways in just a matter of months. Telemedicine, for instance, allowed us to pivot quickly from in-person care to virtual care. We have continued to provide necessary care to our patients while promoting social distancing, reducing the risk of viral spread and recognizing patients’ fears.
Both Cleveland Clinic and Mayo Clinic have gone from providing thousands of virtual visits per month before the pandemic to hundreds of thousands now across a broad range of demographics and conditions. At Cleveland Clinic, 94 percent of diabetes patients were cared for virtually in April.
While virtual visits are here to stay, there are obvious limitations. There is no substitute for in-person care for those who are severely ill or require early interventions for life-threatening conditions. Those are the ones who — even in the midst of this pandemic — must seek the care they need.
Patients who need care at a clinic or hospital or doctor’s office should know they have reduced the risk of Covid-19 through proven infection-control precautions under guidelines from the Centers for Disease Control and Prevention. We’re taking unprecedented actions, such as restricting visiting hours, screening patient and caregiver temperatures at entrances, encouraging employees to work from home whenever possible, providing spaces that allow for social distancing, and requiring proper hand hygiene, cough etiquette and masking.
All of these strategies are intended to significantly reduce risk while allowing for vital, high-quality care for our patients.
The novel coronavirus will not go away soon, but its systemic side effects of fear and deferred care must.
We will continue to give vigilant attention to Covid-19 while urgently addressing the other deadly diseases that haven’t taken a pause during the pandemic. For patients with medical conditions that require in-person care, please allow us to safely care for you — do not delay. Lives depend on it.


India opened up restaurants, shopping malls and places of worship today even as it recorded a record-high 9,971 new coronavirus cases, the third-most worldwide behind Brazil and the U.S.
Why it matters: Lockdowns are being lifted in South Asia — home to one-quarter of the world’s population — not because countries are winning the battle against COVID-19, but because they simply can’t sustain them any longer.
Flashback: For a time, South Asia was cited as a source of optimism because relatively few cases and deaths were being recorded despite large, dense populations.

Limited testing means South Asia’s outbreaks could actually be far more severe. India, for example, is testing at one-twentieth the rate of the U.S.
Bhramar Mukherjee, a professor at the University of Michigan who has been modeling India’s outbreak, tells Axios that while some states have hit initial peaks, she doesn’t expect a national peak until late July or August.
Zoom in: Mumbai has launched an app to help people locate hospitals with empty beds, but such is the scarcity that they’re often full by the time patients arrive, WSJ reports. Some die without ever receiving treatment.
The coronavirus likely arrived in Mumbai with wealthy people returning from abroad, before spreading among poorer people and to slums where social distancing is hardly an option.
The bottom line: South Asian governments attempted to balance health and hunger, knowing they could only shut down their largely informal economies for so long.
https://www.axios.com/sports-betting-stock-market-surge-0e945773-d676-4f0a-a6a0-a0f92611b10b.html
Professional investors have largely abandoned the stock market amid the coronavirus pandemic, but sports bettors and bored millennials have jumped into the retail stock trading market with both feet.
Why it matters: They may be a driving force pushing U.S. stocks to their recent highs — and potentially driving them further.
What’s happening: Online brokerages have seen a record number of new accounts opened this year, and the big four — E-Trade, TD Ameritrade, Charles Schwab and Interactive Brokers — executed as many trades in March and April as in the whole first half of last year, per public disclosures.
Between the lines: Robinhood, whose easy-to-use app makes the transition between sports betting and trading seamless, boasts a similar customer base to most sportsbooks, notes Marc Rubinstein in his newsletter, Net Interest.
The big picture: Sports betting and stock trading aren’t all that different. In fact, most online betting platforms are modeled on stock exchanges, and Nasdaq itself provides sportsbooks with technology that was born in the financial markets.
Meanwhile, most professional investors were sitting on the sidelines.
Professionals have also been buying bonds rather than stocks as U.S. equity indexes raced back from their lows over the last two months.
The bottom line: Day trading has replaced sports betting as a form of entertainment for many Americans during the shutdown, and this phenomenon could partly explain the current disconnect between the economy (down) and the stock market (up).
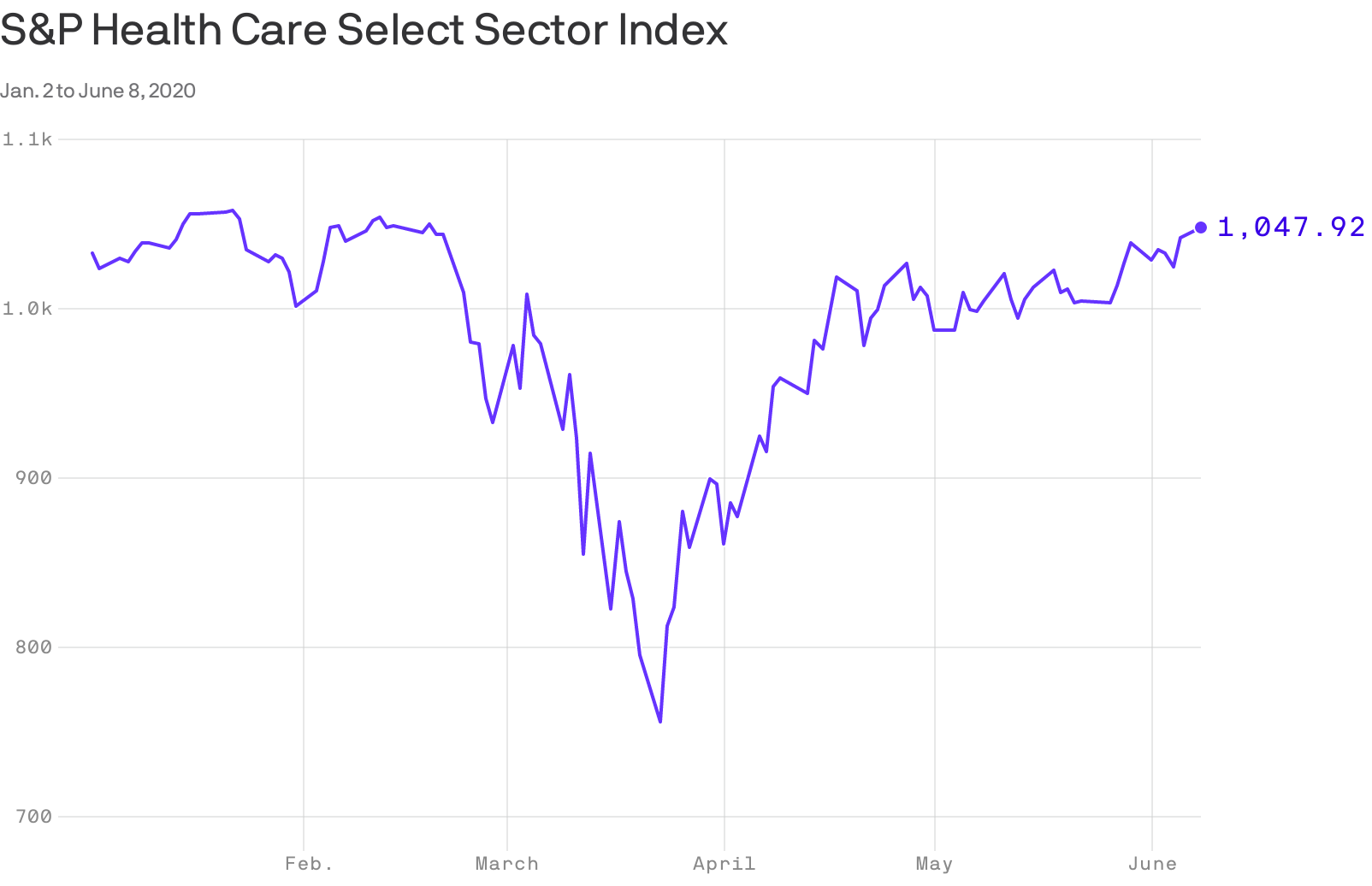
The S&P index of top health care companies finished Monday higher than where it opened the year, Axios’ Bob Herman reports.
The big picture: A global coronavirus pandemic, social unrest, mass unemployment, and the halting of medical procedures haven’t been enough to derail Wall Street’s rosy view of the health care industry.
Where things stand: The coronavirus started to affect the economy toward the tail end of the first quarter, but the health care industry was relatively unscathed.
Between the lines: The second quarter likely will be worse, as the brunt of the coronavirus lockdown was felt in April and May. But normal operations have already started resuming for some health care sectors, regardless of the virus’ spread.